Research Articles
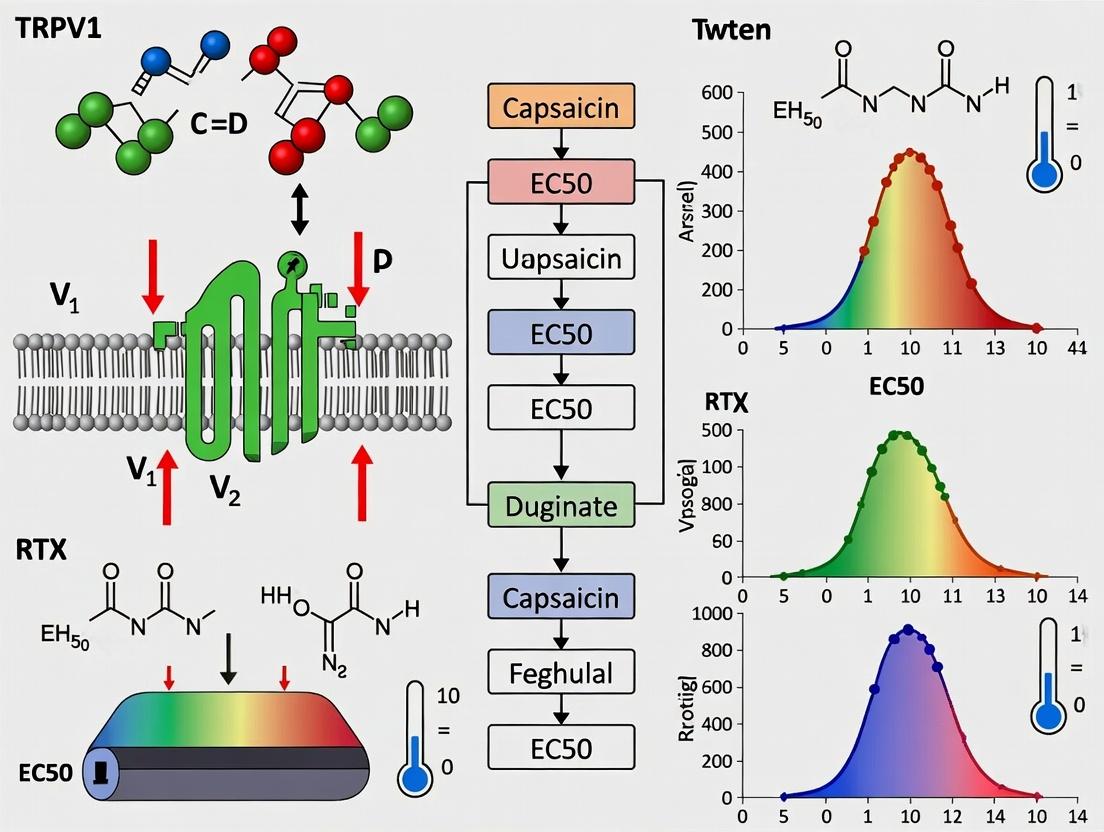
Mastering TRPV1 Channel Pharmacology: A Comprehensive Guide to Automated Patch Clamp Protocol Optimization for Drug Discovery
This article provides a detailed, step-by-step guide for researchers and drug development professionals to optimize automated patch clamp (APC) protocols for studying the Transient Receptor Potential Vanilloid 1 (TRPV1) channel.
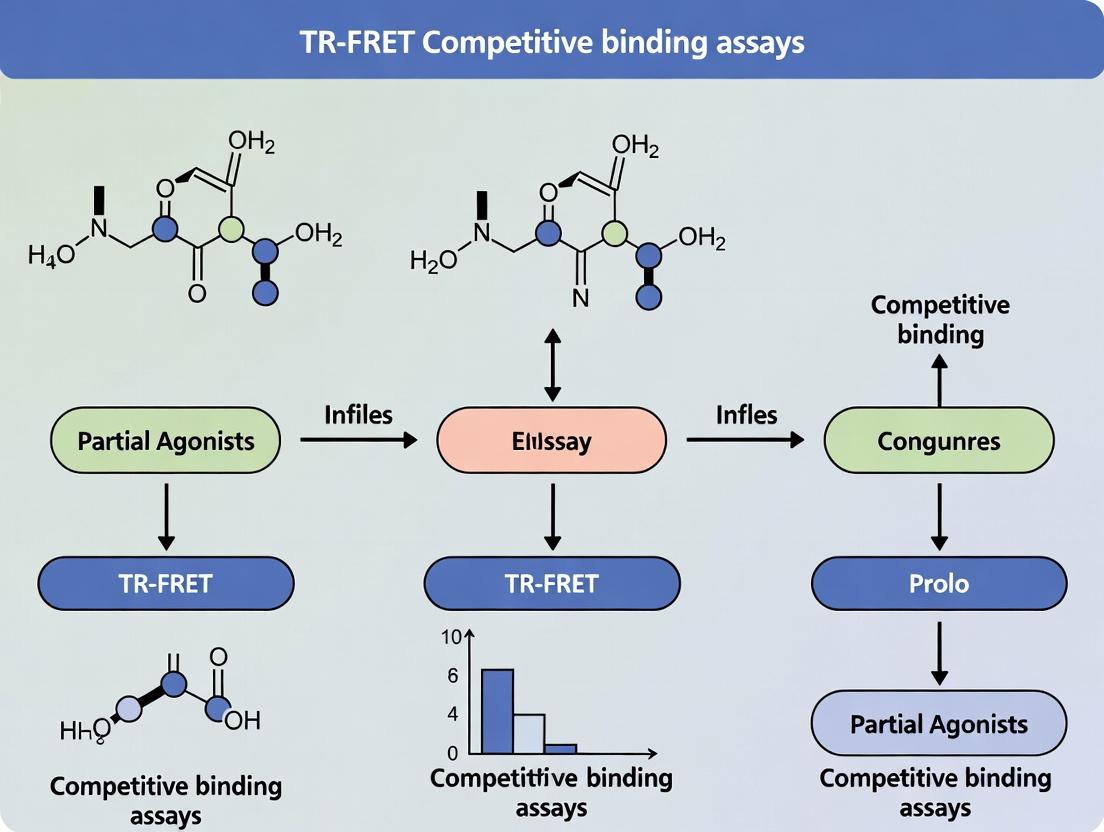
TR-FRET Competitive Binding Assays: A Complete Guide for Profiling Partial Agonists in Drug Discovery
This comprehensive guide details the application of Time-Resolved Förster Resonance Energy Transfer (TR-FRET) competitive binding assays for the precise characterization of partial agonists.
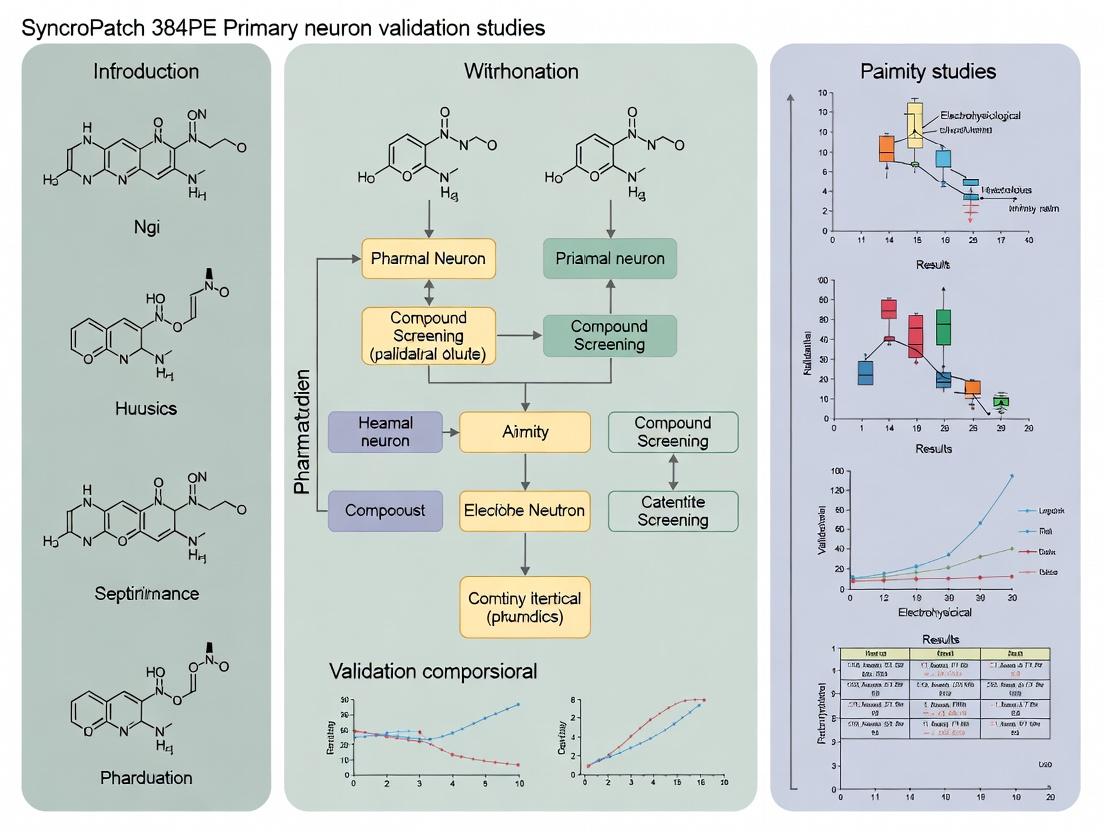
Validating Synaptic Targets: A Comprehensive Guide to SyncroPatch 384PE Studies in Primary Neurons
This article provides researchers, scientists, and drug development professionals with a detailed framework for designing, executing, and interpreting validation studies of the SyncroPatch 384PE automated patch clamp system using primary...
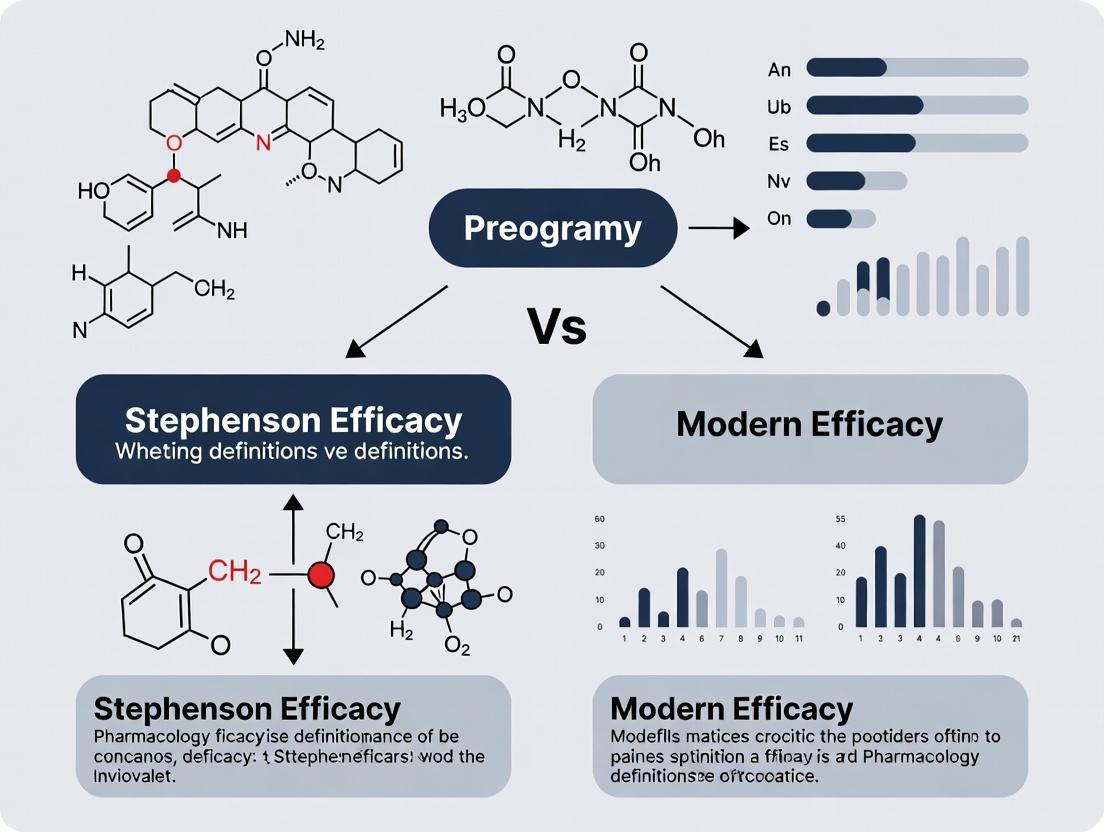
Efficacy Reimagined: Deconstructing the Stephenson vs. Modern Paradigms in Quantitative Pharmacology
This article provides a critical examination for researchers and drug development professionals of the historical and contemporary definitions of pharmacological efficacy.
Solutol Method vs. Pre-Saturation: A Comprehensive Guide to Determining Unbound Fraction (fu) in Drug Discovery
This article provides a detailed comparative analysis of two prominent in vitro methods for determining the unbound drug fraction (fu): the Solutol (co-solvent) method and the Pre-saturation technique.
Mastering Schild Analysis: A Step-by-Step Guide for Determining Antagonist Affinity and pA₂
This comprehensive guide for researchers and drug development professionals provides a complete framework for Schild analysis, the gold-standard method for quantifying antagonist affinity (pA₂/pK<sub>B</sub>) at G-protein coupled receptors (GPCRs) and...
SPA vs Filtration Binding Assays: A Comprehensive Guide for Drug Discovery Researchers
This article provides a detailed comparison of Scintillation Proximity Assays (SPA) and traditional filtration-based binding assays for researchers in drug discovery.
SPA Bead Selection Guide: Strategies for Achieving Ultra-Low Background in Scintillation Proximity Assays
This comprehensive guide details the critical process of selecting scintillation proximity assay (SPA) beads to minimize non-specific background signal, a major challenge in high-throughput screening and quantitative bioassays.
SNAC vs. Sodium Caprylate: Mechanisms and Applications as Oral Drug Absorption Enhancers
This article provides a comprehensive analysis of two prominent pharmaceutical absorption enhancers, SNAC (Salcaprozate sodium) and sodium caprylate.
SMAD Transcription Factors: The Central Signaling Hub of the TGF-β Pathway in Health and Disease
This comprehensive review explores the critical role of SMAD transcription factors as the primary intracellular effectors of the TGF-β signaling superfamily.