ICH Q1B Photostability Testing Explained: Strategic Application of Forced Degradation vs. Confirmatory Studies in Drug Development
This article provides a comprehensive guide to ICH Q1B photostability testing for drug development professionals, differentiating between forced degradation (stress) and confirmatory studies.
ICH Q1B Photostability Testing Explained: Strategic Application of Forced Degradation vs. Confirmatory Studies in Drug Development
Abstract
This article provides a comprehensive guide to ICH Q1B photostability testing for drug development professionals, differentiating between forced degradation (stress) and confirmatory studies. It covers foundational principles, methodological execution, and troubleshooting strategies, concluding with a comparative analysis to validate product stability and packaging decisions. The content synthesizes current regulatory expectations with practical application to ensure robust drug product development and regulatory compliance.
Demystifying ICH Q1B: Core Principles and the Critical Distinction Between Forced Degradation and Confirmatory Testing
ICH Q1B, titled "Photostability Testing of New Drug Substances and Products," is an integral annex to the core stability guideline, ICH Q1A. Its primary purpose is to define the basic principles for conducting photostability testing as part of the stress testing of a new drug substance and product to elucidate its inherent photosensitivity. The scope is specific: it provides a standardized approach for evaluating the effects of light, typically from qualified light sources, to ensure drug quality, safety, and efficacy are maintained when the product is exposed to light during manufacturing, packaging, storage, and patient use.
Within the broader thesis contrasting forced degradation studies (development-phase, exploratory) with confirmatory studies (formal, for registration), ICH Q1B primarily defines the protocol for confirmatory studies. These are standardized tests conducted on final drug substances and products in their final market packaging to establish re-test periods, shelf lives, and labeling requirements. Forced degradation studies, while referenced as valuable precursors, are scoped under development and are not standardized by Q1B.
Core Quantitative Data: Light Exposure Conditions
The guideline specifies precise light exposure requirements. The summary of quantitative conditions is presented below.
Table 1: ICH Q1B Confirmatory Testing Light Exposure Conditions
| Condition | Exposure Level | Purpose |
|---|---|---|
| Option 1: Cool White Fluorescent | Minimum 1.2 million lux hours | Simulates overall visible light exposure. |
| Option 1: Near UV Lamp | Minimum 200 watt hours/square meter | Simulates UV light exposure critical for photodegradation. |
| Option 2: Combined Source (e.g., Xenon, Metal Halide) | Exposure must meet both the lux hour and watt hour/square meter criteria above. | A single, balanced source providing full spectral output. |
Detailed Experimental Protocols
Protocol 1: Confirmatory Testing for Drug Substance
Objective: To evaluate the photosensitivity of the drug substance itself.
- Sample Preparation: Place a sufficient quantity of drug substance in a suitable, inert, transparent container (e.g., clear glass or plastic dish). The sample thickness should not exceed 3 mm (a typical monolayer of powder).
- Analysis: Forced degradation samples may be used for method development, but formal confirmatory samples are analyzed.
- Procedure: Expose the sample to the total light dose specified in Table 1 (Option 1 or 2). A protected control sample (wrapped in aluminum foil) must be stored under identical temperature conditions.
- Analysis: Post-exposure, analyze both exposed and control samples using validated stability-indicating methods (e.g., HPLC). Compare for changes in appearance, assay, and degradation products.
- Decision Flow: If the drug substance shows significant change, it is classified as photosensitive, and protective packaging is required. The drug product must then be tested.
Protocol 2: Confirmatory Testing for Drug Product
Objective: To assess the photostability of the marketed product in its immediate pack.
- Sample Preparation: Test at least three primary batches of the final product in its proposed market packaging (immediate container/closure system).
- Procedure: Expose samples to the full light dose as per Table 1. A protected control sample is mandatory.
- Analysis: Analyze exposed and control samples for critical quality attributes: appearance, assay, degradation products, dissolution (for solids), and pH (for liquids).
- Decision Flow:
- If the product is acceptable (meets all specifications), no further testing is needed.
- If the product shows unacceptable change, testing should be repeated on samples in secondary packaging (e.g., carton).
- If still unacceptable, the product is labeled for special storage conditions (e.g., "Protect from light").
Visualizing the ICH Q1B Testing Logic & Workflow
Title: ICH Q1B Confirmatory Testing Decision Logic
Title: Forced Degradation vs. Confirmatory Studies Scope
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for ICH Q1B Photostability Testing
| Item / Reagent Solution | Function in Photostability Testing |
|---|---|
| Qualified Light Cabinet | Provides controlled, calibrated exposure to cool white fluorescent and near-UV light (or a combined source) meeting ICH Q1B spectral distribution and intensity requirements. |
| Lux Meter & UV Radiometer | Essential for calibrating and confirming the light cabinet delivers the required 1.2 million lux hours and 200 W·hr/m² doses. |
| Validated Stability-Indicating HPLC/UPLC Method | The primary analytical tool for quantifying the active pharmaceutical ingredient (API) and resolving, identifying, and quantifying photodegradation products. |
| Photosensitivity Reference Standards (e.g., Quinine Actinometer) | Chemical systems used to confirm and calibrate the photolytic energy output of the light cabinet, ensuring inter-laboratory reproducibility. |
| Appropriate Inert Sample Containers (e.g., Quartz, Borosilicate Glass Dishes) | Used for drug substance testing; must be transparent to the full spectrum of light used and chemically inert to prevent interaction. |
| Primary and Secondary Market Packaging | The final, commercial container/closure system (e.g., blister packs, vials, bottles) and any secondary carton, which are the actual test articles for drug product confirmatory studies. |
| Protected Control Samples (Aluminum Foil Wrapped) | Crucial for differentiating photolytic change from thermal or other degradation during the test period. Stored alongside exposed samples. |
| Forced Degradation Sample Set | Pre-exposed, highly degraded samples of drug substance/product used during method development to demonstrate the stability-indicating capability of the analytical method. |
1. Introduction: Within the Framework of Forced Degradation vs. Confirmatory Studies
The ICH Q1B guideline, "Photostability Testing of New Drug Substances and Products," establishes a two-tiered approach to light stability evaluation. This framework is built upon a fundamental distinction between forced degradation studies (stress testing) and confirmatory studies (formal stability testing).
- Forced Degradation Studies: These are investigative, research-driven experiments designed to elucidate the intrinsic photostability of a drug substance. They employ extreme conditions of irradiation (e.g., prolonged exposure, high intensity) to identify degradation pathways, characterize degradation products, and validate analytical methods. The batch used is typically a small, non-GMP R&D batch, and the protocol is highly flexible.
- Confirmatory Studies: These are definitive, standardized tests conducted on the final formulation(s) packaged in the proposed market packaging. The primary goal is to provide evidence for the product's label storage instructions and establish its re-test period/shelf life under specific lighting conditions. These studies are performed on Standardized Photostability Batches under strictly controlled, ICH-defined conditions.
This whitepaper focuses on the design, execution, and regulatory significance of the confirmatory study, specifically detailing the requirements for the standardized batches used in registration dossiers.
2. The Standardized Confirmatory Study: Core Principles
The confirmatory study is a formal part of the product stability program. Its design is directly linked to the conditions defined in ICH Q1B and the parent guideline ICH Q1A(R2). The core principle is to test a product under conditions that simulate, in a standardized and accelerated manner, the light exposure it may encounter during storage and use.
2.1. Quantitative Requirements for Light Exposure
ICH Q1B specifies a standardized exposure level. The guideline mandates that samples be exposed to not less than 1.2 million lux hours of visible light and 200 watt hours/square meter of near-UV energy (320-400 nm). The following table summarizes the standard options.
Table 1: Standardized Light Exposure Conditions for Confirmatory Studies
| Light Source | Minimum Exposure Requirements | Typical Achievement Time | Primary Purpose |
|---|---|---|---|
| Cool White Fluorescent (Visible) | ≥ 1.2 million lux hours | ~5-7 days in a calibrated cabinet | Simulates indoor/retail lighting exposure. |
| Near-UV Fluorescent (UV-A) | ≥ 200 Wh/m² (320-400 nm) | ~1-2 days in a calibrated cabinet | Simulates the UV component of daylight. |
2.2. The Standardized Photostability Batch: Key Characteristics
The batch used for the formal confirmatory study is distinct from research batches. Its attributes are summarized below.
Table 2: Characteristics of Standardized Confirmatory vs. Research Forced Degradation Batches
| Characteristic | Confirmatory Study Batch | Forced Degradation Batch |
|---|---|---|
| Purpose | Formal registration; shelf-life justification. | Method development; pathway elucidation. |
| Batch Origin | Primary stability batches (as per ICH Q1A). | Small-scale R&D batches. |
| GMP Status | GMP-manufactured. | Non-GMP. |
| Formulation | Final market formulation(s). | Drug substance or early prototypes. |
| Packaging | In final proposed market container/closure. | Typically unprotected or in clear glass. |
| Protocol | Fixed, ICH Q1B conditions. | Flexible, scientifically justified conditions. |
| Analysis | Validated stability-indicating methods. | Research or in-development methods. |
3. Experimental Protocol for a Standardized Confirmatory Study
The following is a detailed methodology for executing a GMP-compliant confirmatory photostability study.
1. Sample Selection & Preparation:
- Select samples from at least one primary stability batch of the drug product (minimum pilot scale).
- Prepare a minimum of three sets of samples: one for visible light, one for near-UV light, and one protected dark control (wrapped in aluminum foil).
- For combination products, test each component separately if physically possible.
2. Calibration & Chamber Qualification:
- Use a calibrated photostability chamber that meets ICH spectral power distribution requirements.
- Confirm calibration of radiometers (for UV) and lux meters (for visible light) prior to study initiation.
- Perform temperature mapping to ensure uniformity (typically 25°C ±2°C is maintained).
3. Sample Placement & Exposure:
- Place samples and dark controls in the chamber. Ensure samples are positioned to receive uniform illumination.
- Expose one set to the required visible light exposure (≥1.2 million lux hours).
- Expose a separate, identical set to the required near-UV energy (≥200 Wh/m²).
- Monitor cumulative exposure continuously. The "Option 2" integrated approach (simultaneous exposure to both) is also acceptable if the chamber meets both requirements.
4. Post-Exposure Analysis:
- At the conclusion of exposure, retrieve all samples (exposed and dark controls) simultaneously.
- Analyze samples using validated stability-indicating analytical methods (e.g., HPLC/related substances, appearance, assay, dissolution for solid oral dosages).
- Compare results from light-exposed samples against the protected dark controls to isolate the effect of light.
5. Data Interpretation & Reporting:
- Significant change is defined as a failure to meet acceptance criteria (e.g., increase in degradation products, loss of assay, change in physical attributes like color or dissolution).
- Results directly inform storage statements on the label ("Protect from light" if failing, or no statement if passing).
4. Visualizing the ICH Q1B Decision Pathway
The following diagram illustrates the logical decision-making process for photostability testing as per ICH Q1B, highlighting the role of the confirmatory study.
Title: ICH Q1B Photostability Testing Decision Pathway
5. The Scientist's Toolkit: Key Research Reagent Solutions for Photostability Studies
Table 3: Essential Materials and Equipment for ICH Q1B Confirmatory Studies
| Item / Solution | Function / Purpose | Key Specifications / Notes |
|---|---|---|
| ICH-Compliant Photostability Chamber | Provides controlled exposure to visible and UV light as per ICH Q1B spectral requirements. | Must be qualified and calibrated for irradiance (W/m²) and illuminance (lux) uniformity. |
| Calibrated Radiometer & Lux Meter | Measures cumulative UV energy (Wh/m²) and visible light intensity (lux hours). | Calibration traceable to national standards; used for chamber validation and study monitoring. |
| Validated Stability-Indicating HPLC/UPLC Method | Quantifies drug substance and identifies/dequantifies photodegradation products. | Must be validated for specificity, precision, accuracy, and robustness per ICH Q2(R1). |
| Chemical Actinometers | Used for secondary verification of light dose in forced degradation studies (e.g., quinine monohydrochloride). | Not typically required for standardized confirmatory studies but crucial for method development. |
| Light-Opaque Control Containers | (e.g., aluminum foil, amber film) To wrap dark control samples. | Must provide complete protection from light to serve as a valid baseline for comparison. |
| GMP-Manufactured Drug Product Batch | The test article for the formal confirmatory study. | Must be from a primary stability batch, representing the final formulation and manufacturing process. |
| Proposed Market Packaging | The container-closure system in which the product is tested. | Testing is performed on the product in its immediate pack; secondary pack may also be evaluated. |
Forced degradation, or stress testing, is an essential development tool used to elucidate degradation pathways and establish the intrinsic stability of a drug substance. This technical guide frames the practice within the critical distinction between forced degradation studies (development, mechanistic) and confirmatory stability studies (regulatory, ICH-guided). A core thesis is that while ICH Q1B photostability testing provides a standardized confirmatory protocol to validate the stability of a marketed product under specific light conditions, forced degradation is a development tool. The latter employs more severe, non-physiological stress conditions (e.g., harsher light, extreme pH, high temperature, oxidation) to proactively identify potential degradation products, elucidate pathways, and validate analytical methods. Forced degradation informs the design of confirmatory studies like ICH Q1B but serves a fundamentally different, upstream purpose in the drug development lifecycle.
Core Objectives and Stress Conditions
The primary objectives of forced degradation studies are:
- To identify potential degradation products and pathways.
- To determine the intrinsic stability of the molecule.
- To validate the specificity and stability-indicating capability of analytical methods.
- To provide insights for formulation development and packaging selection.
Standard stress conditions are summarized in Table 1.
Table 1: Standard Forced Degradation Stress Conditions and Typical Protocols
| Stress Condition | Typical Protocol Parameters | Target Degradation (%) | Key Degradation Pathways Elucidated |
|---|---|---|---|
| Acidic Hydrolysis | 0.1–1 M HCl, 25–70°C, 1–7 days | 5-20% | Hydrolysis (e.g., amide, ester, lactam), rearrangement. |
| Basic Hydrolysis | 0.1–1 M NaOH, 25–70°C, 1–7 days | 5-20% | Hydrolysis, deamidation, β-elimination, racemization. |
| Oxidative Stress | 0.1–3% H₂O₂, room temperature, hrs-1 day | 5-20% | Oxidation of methionine, cysteine, tryptophan; N-oxide formation. |
| Thermal Stress (Solid) | 50–80°C, dry oven, 1–4 weeks | <10% | Dehydration, pyrolysis, cyclization, solid-state interactions. |
| Thermal Stress (Solution) | 40–80°C, pH-dependent, 1–7 days | 5-20% | Hydrolysis, oxidation, aggregation. |
| Photostress (Forced) | >1.2 million lux hours visible & 200 W·h/m² UVA (often exceeded) | ~10% | Photolysis, radical-mediated oxidation, ring rearrangements. |
| Humidity Stress | 75–90% Relative Humidity, 25–40°C, 1–4 weeks | <10% | Hydrolysis, hydrate formation, physical changes. |
Detailed Experimental Protocol: Forced Photodegradation vs. ICH Q1B
A comparative protocol highlights the distinction between development and confirmatory studies.
Protocol: Forced Photodegradation (Mechanistic Elucidation)
- Objective: To identify all possible photodegradants and pathways.
- Sample Preparation: Prepare drug substance and product solutions in multiple solvents (water, methanol, pH buffers) and solid-state samples. Use quartz vials for solutions to allow full UV transmission.
- Light Source: A combination of cool white fluorescent (emitting ~UV/visible) and near-UV fluorescent (UVA, 320-400 nm) lamps. Xenon or metal halide lamps may be used for higher intensity.
- Stress Conditions: Samples are exposed to significantly higher total illumination than ICH Q1B—often 2-5 times the minimum standard (e.g., 600 W·h/m² UVA). Samples may be exposed in both powdered and compacted forms. Intermittent irradiation cycles (e.g., 24 hrs on/off) may be used.
- Analysis Timepoints: Multiple timepoints (e.g., 0, 24, 48, 96, 200+ W·h/m²) to monitor progression.
- Analysis: HPLC-UV/PDA, LC-MS/MS, SFC-MS for identification; NMR for structural elucidation.
Protocol: ICH Q1B Confirmatory Photostability Testing
- Objective: To confirm that standard packaging provides adequate protection under typical handling and storage conditions.
- Sample Preparation: Drug product in its immediate primary pack, and drug substance as per standard batch quality.
- Light Source: As per Option 1 or 2 in ICH Q1B (specific cool white and near-UV fluorescent lamps).
- Stress Conditions: Minimum exposure of 1.2 million lux hours of visible light and 200 W·h/m² of UVA energy. A single exposure point is standard.
- Analysis Timepoints: Single point analysis after full exposure.
- Analysis: HPLC-UV comparison against protected control; focus on meeting acceptance criteria (e.g., related substances).
Elucidating Degradation Pathways: A Logical Workflow
Forced degradation data is used to construct degradation pathways. The following diagram illustrates the logical workflow from stress to pathway elucidation.
Title: Forced Degradation Pathway Elucidation Workflow
A generalized oxidative degradation pathway for a model molecule containing sulfide and phenol moieties is shown below.
Title: Example Oxidative/Photolytic Degradation Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials and Reagents for Forced Degradation Studies
| Item / Reagent Solution | Function in Forced Degradation Studies |
|---|---|
| Controlled Stability Chambers (e.g., Thermostatic Ovens, Humidity Chambers) | Provide precise, reproducible temperature and relative humidity conditions for thermal and humidity stress studies. |
| Photostability Chambers (ICH-Q1B compliant & extended intensity) | Provide calibrated light exposure for both confirmatory (ICH) and more severe forced photodegradation studies. |
| High-Purity Acids & Bases (e.g., HCl, NaOH, Trifluoroacetic Acid) | Used to prepare stress solutions for hydrolytic degradation studies under acidic and basic conditions. |
| Oxidizing Agents (e.g., Hydrogen Peroxide (H₂O₂), Azo Initiators like AIBN, Metal Salts) | Induce oxidative degradation pathways. H₂O₂ is most common; radical initiators explore different mechanisms. |
| Deuterated Solvents (e.g., D₂O, CD₃OD) | Used for preparing samples for NMR analysis to elucidate degradant structures. |
| LC-MS Grade Solvents & Buffers | Essential for high-performance liquid chromatography (HPLC) and mass spectrometry (MS) analysis to prevent artifact generation. |
| Stability-Indicating HPLC Columns (C18, Phenyl, HILIC) | Different chromatographic phases are used to separate and resolve a wide range of potential degradants from the API. |
| Chemical Traps / Scavengers (e.g., Methionine, Ascorbic Acid, DTT) | Used in mechanistic studies to quench specific reactive species (e.g., radicals) and confirm proposed pathways. |
In the framework of ICH Q1B photostability testing, a critical distinction governs strategic planning: forced degradation (stress testing) and confirmatory (formal stability) studies serve two divergent primary objectives. Forced degradation is an investigative tool for Method Development & Pathway Identification, aiming to elucidate degradation chemistry and validate analytical methods. Confirmatory studies under ICH Q1B are exercises in Product Qualification, intended to verify that a product remains within acceptance criteria under specified light conditions. This guide delineates the experimental and philosophical separation of these objectives, which, while interconnected, demand distinct protocols, acceptance criteria, and data interpretation.
Core Objectives: A Comparative Framework
Table 1: Primary Objectives and Characteristics
| Aspect | Product Qualification (Confirmatory ICH Q1B) | Method Development & Pathway Identification (Forced Degradation) |
|---|---|---|
| Primary Goal | Confirm package suitability & product stability under labeled storage conditions. | Identify degradation pathways, develop & validate stability-indicating methods. |
| Regulatory Driver | ICH Q1B (Photostability Testing of New Drug Substances and Products). | ICH Q2(R2) / Q14 (Analytical Procedure Development), ICH Q1A (Stability). |
| Sample State | Final marketed package (primary & secondary). | Often unpackaged drug substance/product, or product in transparent container. |
| Stress Intensity | Controlled, defined (e.g., 1.2 million lux hours, 200 W h/m² UVA). | Exaggerated, non-linear (e.g., extended UV exposure, higher temperature/humidity). |
| Acceptance Criteria | Pre-defined specifications (e.g., assay, impurities, appearance). | No product acceptance criteria; goal is sufficient degradation (~5-20%). |
| Key Output | Evidence for product labeling and shelf-life. | Degradation profile, elucidated structures, validated analytical method. |
Experimental Protocols
Protocol for Product Qualification (Confirmatory ICH Q1B)
Objective: To demonstrate the product in its proposed packaging can withstand light exposure during storage and use.
- Sample Preparation: Use final drug product in its immediate primary pack (e.g., blister, bottle). Include secondary packaging if opaque. Prepare duplicate sets: one for light, one for dark control (wrapped in aluminum foil).
- Light Exposure: Expose samples per ICH Q1B Option 1 or Option 2.
- Option 1: Sample exposed to 1.2 million lux hours of visible light and 200 watt-hours/square meter of near-ultraviolet (UVA, 320-400 nm).
- Option 2: Sample exposed to cool white fluorescent and UVA lamps to meet the same integrated limits.
- Analysis: Analyze light-exposed and dark control samples for:
- Appearance (color, clarity, dissolution for solids).
- Potency (assay).
- Degradation products (related substances test).
- Acceptance: Product meets its established specification. Any change is reported and justified.
Protocol for Method Development & Pathway Identification (Forced Degradation)
Objective: To deliberately degrade the sample to identify likely degradation products and pathways, ensuring the analytical method can separate and detect them.
- Sample Preparation: Use drug substance (powder) or product (without primary pack or in clear glass). Prepare solutions and/or solid-state samples.
- Stress Conditions (not linear; sample-dependent):
- Photolysis: Exposure to light significantly exceeding ICH Q1B limits (e.g., up to 2000 W h/m² UVA). May use different light sources (e.g., UVB, simulated sunlight).
- Heat/Humidity: e.g., 60°C/75% RH for solids; 40-70°C in solution at various pH.
- Oxidation: Exposure to 0.1-3% H₂O₂ or AIBN.
- Hydrolysis: Acid/Base treatment (e.g., 0.1M HCl/NaOH) at elevated temperature.
- Analysis: Use hyphenated techniques (LC-MS/MS, LC-TOF) to:
- Monitor degradation over time.
- Isolate and characterize major degradation products (>0.1%).
- Establish mass balance.
- Output: Degradation pathway map, identification of degradation products, and validation that the analytical method is stability-indicating (resolves all degradants from analyte).
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Materials for Photostability and Forced Degradation Studies
| Item | Function in Qualification | Function in Method Development |
|---|---|---|
| ICH-Q1B Compliant Light Cabinet | Provides controlled, calibrated exposure per ICH Option 1/2. | Used for baseline studies; often supplemented with other sources. |
| High-Intensity UV Light Source (e.g., Xenon arc) | Not typically used. | Provides exaggerated photolytic stress for faster pathway identification. |
| LC-MS/MS System with PDA | For routine impurity profiling per validated method. | Critical for separation, detection, and structural elucidation of novel degradants. |
| Stability Chamber (Temp/Humidity) | For dark control storage. | For conducting thermal/humidity stress in parallel. |
| Chemical Stressors (H₂O₂, HCl, NaOH) | Not used. | Used to induce hydrolytic and oxidative degradation pathways. |
| Reference Standards of Known Degradants | Used for quantification in confirmatory testing. | Used to confirm degradation pathways and method selectivity. |
Data Presentation: Comparative Outcomes
Table 3: Typical Data Outputs and Interpretation
| Metric | Product Qualification Outcome | Method Development Outcome |
|---|---|---|
| % Potency Loss | Must be within specification (e.g., 95-105%). May be minimal (<2%). | Not applicable for product acceptance. 5-20% loss is often targeted. |
| New Degradant Level | Must be below reporting/threshold limits (e.g., <0.10%). | Identified and characterized, regardless of level (could be >1%). |
| Mass Balance | Calculated to support assay validity. | Critical Requirement. Must be ~98-102% to prove method's stability-indicating capability. |
| Conclusion Statement | "The product, in the proposed packaging, meets ICH Q1B requirements." | "Five major degradants were identified. The HPLC method successfully resolves all from the API and is suitable for stability testing." |
Visualizing the Strategic Pathway
Title: Forced Degradation Informs Confirmatory Study Design
Title: Degradation Pathway Identification Informs Method Development
The Common Technical Document (CTD) is the standardized format for regulatory submissions to the ICH regions. This guide delineates where various study types, including photostability studies, are positioned within the CTD modules, with a specific focus on the distinction between forced degradation (supportive development research) and confirmatory (regulatory) stability studies. This is framed within the broader thesis on ICH Q1B photostability testing, which necessitates both types of studies: forced degradation to understand the molecule's intrinsic photosensitivity and confirmatory studies to define the retest period or shelf life under specific packaging/storage conditions.
The CTD Structure and Study Placement
The CTD is organized into five modules. Modules 2, 3, 4, and 5 contain the technical reports and data. The placement of a study is dictated by its primary purpose within the drug development and registration paradigm.
Table 1: Primary CTD Modules and Content
| CTD Module | Title | Content Overview | Relevant Study Types |
|---|---|---|---|
| Module 1 | Regional Administrative Information | Not part of the harmonized CTD; contains region-specific forms. | N/A (Non-technical) |
| Module 2 | CTD Summaries | Non-clinical and clinical overviews and summaries. | High-level summaries of stability, photo, and forced degradation data. |
| Module 3 | Quality | Chemical, Pharmaceutical, and Biological Information. | Primary location for all drug substance and product stability data. |
| Module 4 | Non-clinical Study Reports | Toxicology and Pharmacokinetics reports. | Photo-toxicity studies (if applicable). |
| Module 5 | Clinical Study Reports | Human study reports. | In-use stability studies supporting clinical trial protocols. |
Within Module 3, data is further organized into specific sections. Stability data, including photostability, is located in the S.7 (Drug Substance) and P.8 (Drug Product) sections.
Table 2: Detailed Placement of Stability & Related Studies in Module 3
| CTD Section | Sub-section | Typical Content | Study Type Classification |
|---|---|---|---|
| S.7 / P.8 | Stability | Summary & Conclusions | High-level conclusions from confirmatory studies. |
| S.7 / P.8 | Stability | Post-approval Stability Protocol & Commitment | Future confirmatory studies. |
| S.7 / P.8 | Stability | Stability Data | Tabulated results from primary confirmatory long-term, accelerated, and photostability studies. |
| S.3 / P.2 | Pharmaceutical Development | Drug Substance / Product Development | Forced degradation studies (stress testing) to elucidate degradation pathways, validate analytical methods, and support formulation development. |
| S.4 / P.5 | Control of Drug Substance / Product | Justification of Specification | Data from forced degradation used to justify the inclusion (or exclusion) of specific impurities in specifications. |
| S.6 / P.7 | Reference Standards or Materials | Information | Characterization data may reference forced degradation samples. |
Forced Degradation vs. Confirmatory Studies in ICH Q1B Context
The ICH Q1B guideline "Photostability Testing of New Drug Substances and Products" inherently requires an understanding from both development and regulatory perspectives.
- Confirmatory Studies (CTD: Module 3, S.7/P.8): These are formal, GMP-compliant studies conducted on primary batches under defined, ICH-prescribed conditions (e.g., Option 1: 1.2 million lux hours visible light, 200 watt hours/m² UV). Their purpose is to confirm the re-test period/shelf life and labeling storage statements. The results are presented in structured stability data tables.
- Forced Degradation (Supportive Research) (CTD: Module 3, S.3/P.2): These are investigative, non-GMP studies designed to elucidate the intrinsic stability of the molecule. They use more severe conditions (e.g., higher intensity light, extended exposure) than ICH Q1B to force degradation. The goal is to understand degradation pathways, identify potential impurities, and validate the specificity of analytical methods. Results are described in the Pharmaceutical Development section.
Table 3: Comparative Analysis of Forced Degradation vs. Confirmatory Photostability Studies
| Parameter | Forced Degradation (Stress Testing) | Confirmatory ICH Q1B Photostability Study |
|---|---|---|
| Primary Objective | To identify likely degradation products, elucidate pathways, and validate analytical methods. | To provide data for labeling (storage conditions) and confirm shelf-life/re-test period. |
| CTD Location | Module 3, S.3 (Drug Substance Development) or P.2 (Product Development). | Module 3, S.7 (Drug Substance Stability) or P.8 (Drug Product Stability). |
| Batch Selection | Development batches (e.g., synthetic route, formulation prototypes). | Primary batches for registration (same as formal stability). |
| Study Conditions | Exaggerated and not standardized; beyond ICH conditions to ensure degradation. | Strictly defined by ICH Q1B (Option 1 or 2). |
| GMP Compliance | Not required (Development study). | Expected for primary stability batches. |
| Data Presentation | Descriptive narrative with supporting chromatograms/spectra in the development report. | Tabulated quantitative results (e.g., assay, impurities) in stability data tables. |
| Regulatory Role | Supportive, explanatory. | Definitive, registrational. |
Experimental Protocols
Protocol for ICH Q1B Confirmatory Photostability Testing
Objective: To determine the effects of light on the drug substance/product as per ICH Q1B for regulatory submission. Materials: Drug substance/product (primary batch), qualified photostability chamber, calibrated light sources (UV & cool white/ID65), lux meter, UV energy meter, appropriate containers/closures. Procedure:
- Sample Preparation: Prepare a minimum of two sets of samples (drug product in immediate pack, and if necessary, in marketing pack; drug substance as thin layer or in suitable transparent container).
- Calibration: Ensure chamber delivers uniform exposure. Calibrate using a validated chemical actinometer or radiometers/lux meters.
- Exposure (Option 1 - Standard): Expose one set to 1.2 million lux hours of visible light and 200 watt hours/m² of UV (320-400 nm). Maintain controlled temperature (e.g., 25°C).
- Control: Protect the second set (dark control) from light using opaque materials but subject to the same temperature.
- Analysis: After exposure, analyze both exposed and control samples for changes in appearance, assay, degradation products, and other quality attributes.
- Decision Tree: If significant change occurs, proceed to testing with protective packaging to confirm sufficiency.
Protocol for Forced Degradation Photostability (Supportive Research)
Objective: To force photodegradation to evaluate intrinsic photosensitivity, identify degradation products, and validate method specificity. Materials: Drug substance/product (development batch), high-intensity light source (e.g., xenon, metal halide), controlled temperature chamber, analytical equipment (HPLC, LC-MS). Procedure:
- Scouting: Expose samples to a range of intensities and durations, significantly exceeding ICH Q1B conditions (e.g., 3-5x UV energy).
- Monitoring: Withdraw samples at multiple time points (e.g., 24h, 48h, 120h).
- Analysis: Perform comprehensive analysis using stability-indicating methods (HPLC/UV, LC-MS). Monitor for loss of parent compound and formation of new peaks.
- Characterization: Isolate and characterize major degradants (e.g., via LC-MS/MS, NMR) to propose degradation pathways.
- Method Challenge: Inject stressed samples into the proposed routine analytical methods to prove specificity—no co-elution of degradants with the main peak.
Diagram 1: Photostability Study Flow from R&D to CTD (76 chars)
The Scientist's Toolkit: Research Reagent & Material Solutions
Table 4: Essential Materials for Photostability Testing
| Item | Function | Notes for Application |
|---|---|---|
| ICH-Compliant Light Chambers | Provide controlled exposure to UV and visible light as per ICH Q1B Option 1 or 2. | Must be qualified (IQ/OQ/PQ) and calibrated regularly. |
| Chemical Actinometers | Validate the radiant exposure (energy) delivered by the light source. | Quinine hydrochloride is a common solution actinometer for UV. |
| Calibrated Radiometers/Lux Meters | Measure UV irradiance (W/m²) and visible illuminance (lux). | Critical for monitoring and documenting exposure conditions. |
| Transparent Containers | Hold samples during exposure (e.g., quartz, borosilicate glass vials). | Must not filter out relevant wavelengths; validation may be needed. |
| Opaque Wraps/Covers | Prepare protected "dark control" samples. | Aluminum foil or specialized opaque sleeves. |
| High-Intensity Light Sources (e.g., Xenon arc) | Used in forced degradation to induce photodegradation. | Provides a broad-spectrum output simulating sunlight. |
| Stability-Indicating HPLC Columns | Separate and quantify the parent compound and its photodegradants. | Columns should be resistant to mobile phases used for method development. |
| LC-MS/MS Systems | Identify and characterize unknown degradation products formed during forced degradation. | Essential for elucidating degradation pathways in development. |
This whitepaper provides an in-depth technical guide to the core parameters governing pharmaceutical photostability testing, as mandated by ICH Q1B. Within the broader thesis contrasting forced degradation (stress testing to elucidate degradation pathways) and confirmatory (compliance testing to establish shelf-life) studies, a precise understanding and control of light sources, irradiance, and integrated quantities is paramount. These parameters directly influence the extent and relevance of photodegradation, impacting the validity of both research paradigms for drug development professionals.
The choice of light source is fundamental, as it defines the spectral regions (UV and visible) to which a drug substance or product is exposed. ICH Q1B specifies two standard options.
Table 1: Standard ICH Q1B Light Source Options
| Source Type | Description | Key Spectral Output | Typical Application |
|---|---|---|---|
| Option 1 | Cool White Fluorescent & Near-UV Fluorescent | Visible (400-800 nm) & UV (320-400 nm) | Comprehensive testing covering full ICH spectrum. |
| Option 2 | A single lamp mimicking solar spectral distribution (e.g., Xenon, metal halide) | Combined UV/Visible, similar to D65/ID65 | Simulates indoor filtered daylight; often used in controlled chambers. |
Experimental Protocol for Source Verification: Regular calibration using a spectroradiometer is required. The lamp is turned on and allowed to stabilize (typically 30 min). The spectroradiometer's sensor is placed at the sample exposure position, and a full spectral scan (e.g., 300-800 nm) is performed. The resulting Spectral Power Distribution (SPD) curve must be compared against ICH Q1B benchmarks to confirm compliance before any study.
Irradiance: The Rate of Energy Delivery
Irradiance (W/m² or, commonly for specific bands, W/m²/nm) measures the power of incident light per unit area. It is the critical parameter controlling the rate of the photochemical reaction. In confirmatory testing, maintaining irradiance within a controlled range ensures standardized, reproducible challenge. In forced degradation, varying irradiance can help probe kinetic relationships.
Experimental Protocol for Irradiance Measurement: A calibrated radiometer/photometer with appropriate spectral filters (e.g., for UV, UVA, visible bands) is used. The sensor is positioned precisely where the sample will be placed, ensuring it is normal (perpendicular) to the light source. Multiple measurements across the exposure area are taken to map and confirm homogeneity (typically within ±10% of the target). Data is recorded at study initiation and at regular intervals (e.g., daily).
Integrated Quantities: The Total Dose
The photochemical effect is a function of the total photon energy absorbed. Integrated quantities, the product of irradiance and time, define the total light dose delivered. ICH Q1B sets minimum exposure levels in terms of integrated energy.
Table 2: ICH Q1B Minimum Required Integrated Exposure
| Spectral Region | Minimum Integrated Energy |
|---|---|
| Ultraviolet (320-400 nm) | 1.2 million W·hr/m² (or 200 W·hr/m² of near-UV at 320-400 nm) |
| Visible (400-800 nm) | 1.2 million Lux·hours |
Experimental Protocol for Dose Calculation and Control:
- For UV Dose: Measure the average UV irradiance (W/m²) at the sample plane. The exposure time (in hours) required is calculated as: Time (hr) = 1.2 x 10⁶ / Measured UV Irradiance (W/m²).
- For Visible Dose: Measure the average illuminance (Lux). The exposure time required is: Time (hr) = 1.2 x 10⁶ / Measured Illuminance (Lux).
- The exposure is controlled using timers or chamber software, often continuing until the longer of the two calculated times is met. Real-time integrating systems are recommended for highest accuracy.
Title: Photostability Testing Workflow: Confirmatory vs. Forced Degradation
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Materials for Photostability Testing
| Item | Function | Key Consideration |
|---|---|---|
| ICH-Compliant Light Chamber | Provides controlled exposure with Option 1 or 2 light sources, temperature/humidity control. | Chamber calibration and homogeneity validation are critical. |
| Spectroradiometer | Measures the Spectral Power Distribution (SPD) of the light source for verification. | Must be NIST-traceable and cover 300-800 nm range. |
| Calibrated Radiometer/Photometer | Measures irradiance (W/m²) and illuminance (Lux) for dose calculation. | Requires separate detectors/filters for UV and visible bands. |
| Chemical Actinometers (e.g., Quinine, potassium ferrioxalate) | Quantum yield references; validate photon flux and dose independently of radiometry. | Useful for verifying system performance, especially in forced degradation. |
| Optical Filters (e.g., UV cutoff, bandpass) | Isolate specific spectral regions in forced degradation studies to identify damaging wavelengths. | Must have known and sharp spectral characteristics. |
| Stability-Indicating HPLC/UPLC Method | Analyzes and quantifies degradation products post-exposure. | Must be validated to separate and detect all relevant photodegradants. |
The precise definition and control of light sources, irradiance, and integrated quantities form the bedrock of meaningful photostability data. In confirmatory ICH Q1B testing, they ensure a standardized, reproducible challenge for shelf-life determination. In forced degradation research, their systematic variation becomes a powerful tool for deconstructing degradation kinetics and pathways. Mastery of these parameters, supported by rigorous calibration and measurement protocols, enables researchers to generate data that is both compliant and scientifically insightful, bridging the gap between regulatory requirement and mechanistic understanding.
Executing ICH Q1B Studies: Step-by-Step Protocols for Confirmatory and Forced Degradation Testing
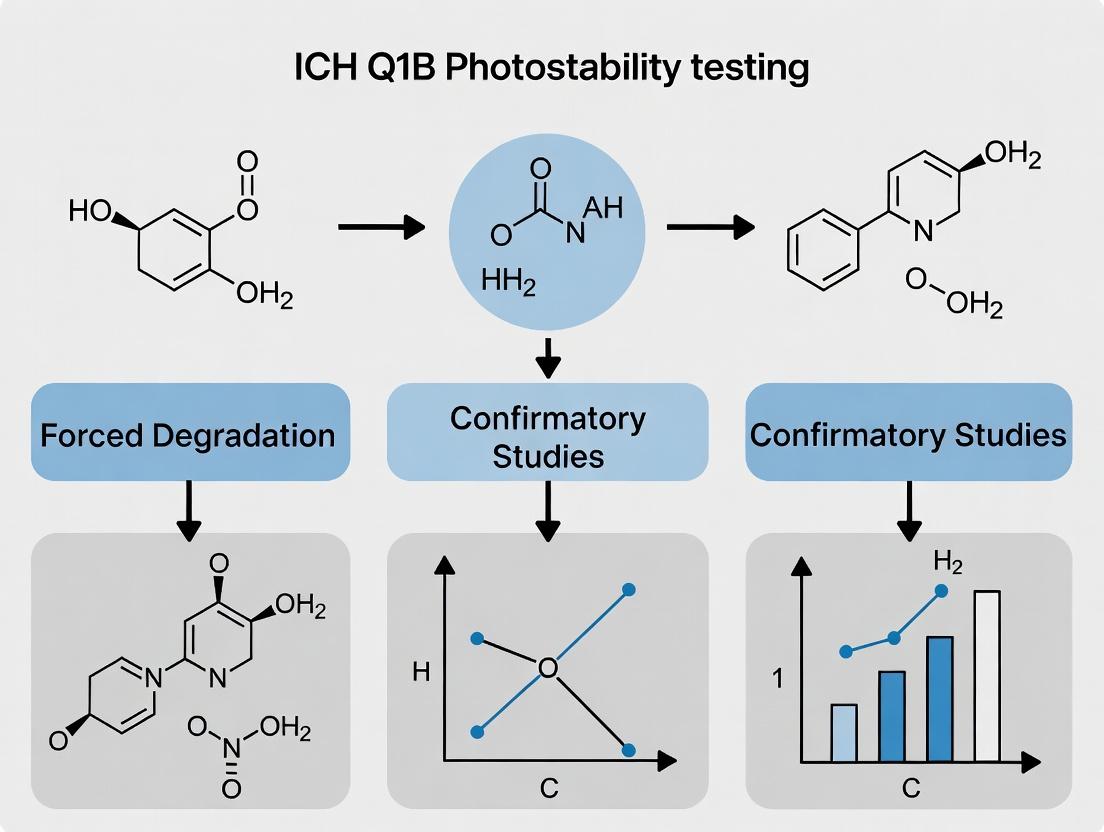
The ICH Q1B guideline, "Photostability Testing of New Drug Substances and Products," provides a framework for assessing the photosensitivity of pharmaceuticals. A critical distinction exists within its methodology: confirmatory testing versus forced degradation studies. Confirmatory tests (Option 1 and Option 2) are formal, standardized photostability studies conducted on a single batch of material under defined conditions to validate the light-protective nature of packaging or to establish a product's light sensitivity for labeling. In contrast, forced degradation studies are stress tests designed to elucidate degradation pathways and establish analytical method stability-indicating power, often using exaggerated conditions. This guide details the core confirmatory testing protocols, focusing on the two primary irradiation strategies, and positions them as essential, regimented components distinct from the investigative nature of forced degradation research.
Core Irradiation Strategies: Option 1 vs. Option 2
The ICH Q1B guideline offers two quantitatively defined strategies for confirmatory testing.
Table 1: Quantitative Comparison of ICH Q1B Confirmatory Irradiation Strategies
| Parameter | Option 1 (Cool White & UV) | Option 2 (Daylight Simulating) |
|---|---|---|
| Primary Light Source | Combination of two separate lamps. | A single, combined lamp. |
| Visible Source | Cool white fluorescent lamp (emitting 400-800 nm). | A lamp whose spectral power distribution matches D65/ID65 standard (indirect daylight). |
| UV Source | Near-UV fluorescent lamp (emitting 320-400 nm with max energy 350-370 nm). | The same lamp must emit integrated near-UV (320-400 nm) energy meeting the required minimum. |
| Minimum Exposure Criteria | Visible: 1.2 million lux hours.UV: 200 watt-hours/square meter. | Must meet the same integrated energy thresholds as Option 1 for both visible (lux-hours) and UV (watt-hours/m²). |
| Primary Advantage | Well-understood, readily available standard lamps. | Simulates full-spectrum daylight more accurately in a single source. |
| Typical Implementation | Separate exposure chambers or banks for visible and UV, or sequential exposure in the same chamber with lamp switching. | Single exposure in a chamber equipped with the appropriate filtered xenon or metal halide lamp. |
Detailed Experimental Protocol
The following methodology is applicable to both drug substances and products.
Material Preparation and Sample Selection
- Batch: A single batch of drug substance (powder) and/or the final marketed product in its immediate primary packaging is used.
- Sample Configuration:
- Drug Substance: Spread as a layer not thicker than 3 mm (e.g., in a glass or quartz dish). Powder may be exposed as-is or protected with a suitable transparent cover (e.g., quartz) if liable to sublimation.
- Drug Product: Exposed in the primary market pack. A duplicate set of samples should be exposed without primary packaging (e.g., tablets in a watch glass) to assess the protective effect of the packaging.
- Controls: Protected controls (wrapped in aluminum foil or stored in the dark) must be maintained under identical temperature conditions.
Irradiation Procedure
The procedure diverges based on the chosen option.
- Calibration: Verify the irradiance (W/m²) of the UV lamp and illuminance (lux) of the visible lamp at the sample position prior to study initiation.
- Calculation: Calculate required exposure time.
- UV Exposure Time (hours) = 200 W·h/m² ÷ Measured UV Irradiance (W/m²).
- Visible Exposure Time (hours) = 1,200,000 lux·h ÷ Measured Illuminance (lux).
- Exposure: Expose samples to either the cool white or the near-UV source first. The order is not specified by ICH.
- Monitoring: Temperature should be controlled (typically not exceeding 40°C). Continuously monitor cumulative exposure using calibrated radiometers/lux meters.
- Completion: Once the minimum exposure for the first source is met, immediately expose the same samples to the second source until its minimum exposure is met.
Protocol for Option 2 (Combined Source)
- Calibration: Verify the spectral power distribution of the source matches D65/ID65 across 300-800 nm. Measure integrated UV irradiance (320-400 nm) and visible illuminance at the sample plane.
- Calculation: Calculate the total exposure time required to meet both criteria. The limiting factor will be the parameter that takes longer to achieve.
- Time to meet UV criteria = 200 ÷ Measured UV Irradiance.
- Time to meet Visible criteria = 1,200,000 ÷ Measured Illuminance.
- Required Exposure Time = The longer of the two calculated times.
- Exposure: Place samples under the single, calibrated source.
- Monitoring: Control temperature and monitor cumulative UV energy and visible light exposure.
- Completion: Terminate exposure once both minimum criteria have been exceeded.
Post-Irradiation Analysis
- Evaluation: Assess samples alongside dark controls for changes in:
- Appearance (color, clarity, physical form).
- Potency via validated stability-indicating assay (e.g., HPLC).
- Degradation Products (increase in specified or unspecified impurities).
- Judgment: The confirmatory test is "passed" if the material, especially in its primary pack, shows no significant change versus the protected control, confirming the packaging is suitable. Significant change under standardized conditions necessitates light-protective labeling.
Visualizations
Diagram 1: ICH Q1B Photostability Testing Decision Pathway
Diagram 2: Option 1 vs Option 2 Experimental Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Confirmatory Photostability Testing
| Item | Function & Specification |
|---|---|
| Calibrated Light Source(s) | Option 1: Separate cool white fluorescent (e.g., ISO 10977:1993 compliant) and near-UV fluorescent lamps. Option 2: Filtered Xenon arc or metal halide lamp with spectral output matching D65/ID65 standard. |
| Calibrated Radiometer/ Photometer | Measures UV irradiance (W/m², 320-400 nm) and visible illuminance (lux) at the sample plane. Must be NIST-traceable for data integrity. |
| Photostability Chamber | An environmental chamber providing controlled temperature (±2°C) and, if needed, humidity, with uniform light distribution and a turntable for sample rotation. |
| Primary Reference Materials | Chemical actinometers (e.g., quinine monohydrochloride dihydrate for UV, benzophenone/benzoic acid actinometry) to validate the radiant energy received by samples. |
| Sample Presentation Accessories | Quartz or borosilicate glass dishes/petri dishes (for substances), neutral transparent supports for product samples, and aluminum foil for dark controls. |
| Stability-Indicating Analytical Methods | Validated HPLC/UPLC methods with photodiode array (PDA) or mass spectrometric (MS) detection to quantify potency loss and identify/degrade degradation products. |
This guide details the critical considerations for sample presentation and positioning during photostability testing, a core component of the ICH Q1B guideline. The principles outlined here serve to bridge the gap between forced degradation studies (a stress testing tool for elucidating degradation pathways) and confirmatory studies (which verify the suitability of the primary packaging under recommended storage conditions). Proper methodology is dosage form-dependent and is essential for generating reproducible, scientifically valid data that accurately informs drug development and regulatory submission.
Core Principles of Sample Presentation in ICH Q1B
The ICH Q1B guideline stipulates that samples should be exposed to light providing an overall illumination of not less than 1.2 million lux hours and an integrated near ultraviolet energy of not less than 200 watt hours/square meter. The sample presentation must ensure uniform irradiation of all samples and, crucially, represent the worst-case scenario for the marketed product configuration.
Key Quantitative Parameters and Tolerances
Table 1: Core Photostability Exposure Conditions and Sample Considerations
| Parameter | ICH Q1B Minimum Requirement | Dosage Form-Specific Consideration |
|---|---|---|
| Total Visible Light | ≥ 1.2 million lux hours | For clear solutions/containers, ensure full penetration. For opaque solids, exposure of all surfaces is critical. |
| Total UV Energy | ≥ 200 W·h/m² | Primary driver for photochemical reactions. Positioning must ensure UV exposure of the most relevant product layer. |
| Sample Positioning | Not specified (driven by uniformity) | Distance from light source, orientation, and arrangement must guarantee all samples receive equivalent, uniform exposure. |
| Sample Configuration | "As marketed" or "worst-case" | Solid dosage units may be spread as a monolayer. Liquids may need to be inverted. Transdermal patches require removal of liner. |
Dosage Form-Specific Methodologies
Solid Oral Dosage Forms (Tablets, Capsules)
- Protocol: A representative number of units (typically 5-12) are placed in a single layer in suitable transparent or inert containers (e.g., Petri dishes). Powders should be spread to a thickness of typically not more than 3 mm. For coated tablets, both the "face" and "edge" orientations may need to be tested to simulate worst-case exposure of any breach in the coating.
- Positioning Rationale: Ensures direct, uniform exposure of the entire surface area of each unit, simulating a scenario where the product is removed from the primary container.
Liquid and Parenteral Dosage Forms (Solutions, Suspensions)
- Protocol: Samples are placed in the final, transparent primary container (e.g., ampoule, vial, bottle). The containers are filled to the maximum nominal volume. Testing is performed both upright and inverted (or on their side) to ensure exposure of the product in contact with the closure system (e.g., stopper, cap liner) and the container wall.
- Positioning Rationale: Critical photodegradants may form at the liquid-air interface or may be catalyzed by interactions with the closure material. Inversion ensures this interface is exposed to light.
Semi-Solid & Topical Dosage Forms (Creams, Ointments, Gels)
- Protocol: The product is evenly spread on an inert, transparent substrate (e.g., glass plate, quartz plate) to a realistic use thickness (e.g., 0.5-1 mm). Alternatively, samples in final packaging (e.g., tubes) are sampled after exposure. For patches, the protective liner is removed prior to exposure.
- Positioning Rationale: Spreading simulates in-use conditions where a thin layer is exposed to ambient light, which is the relevant stress condition.
Complex Dosage Forms (Powders for Reconstitution, Pressurized MDIs)
- Protocol: Requires a two-part study: 1) Exposure of the solid powder in its container (as per solid dosage forms). 2) Exposure of the reconstituted solution or the bulk drug product prior to filling (as per liquid dosage forms).
- Positioning Rationale: Addresses the photostability of both the manufactured state and the ready-to-use state.
Experimental Workflow for Dosage Form-Specific Photostability Testing
Diagram Title: Photostability Testing Workflow by Dosage Form
Relationship to Forced Degradation and Confirmatory Studies
The rigor in sample presentation directly impacts the utility of data for both forced degradation and confirmatory studies. Forced degradation studies use extreme light exposure (often beyond ICH Q1B minimums) on unpackaged drug substance or simple solutions to identify degradation pathways. Confirmatory studies test the final packaged product under ICH Q1B conditions to verify shelf-life claims. The dosage form-specific methodologies described here are essential for the confirmatory study and for any forced degradation work performed on the drug product intermediate.
Diagram Title: Sample Presentation Links Forced and Confirmatory Studies
The Scientist's Toolkit: Key Research Reagent Solutions & Materials
Table 2: Essential Materials for Photostability Sample Preparation and Positioning
| Item / Reagent Solution | Function in Dosage Form-Specific Testing |
|---|---|
| Inert Substrate (Quartz/Glass Plates) | Provides a chemically inert, UV-transparent surface for spreading semi-solid and solid powder samples to a controlled thickness. |
| Calibrated Lux & UV Radiometer | Validates uniform irradiance across the entire sample plane in the photostability chamber, critical for reproducible positioning. |
| Optical Filters (e.g., Window Glass) | Used in optional studies to simulate exposure behind window glass, assessing protection offered by secondary packaging or storage conditions. |
| Inert Sample Containers (Petri Dishes, Watch Glasses) | Holds solid dosage units or powder samples during exposure without interacting photochemically with the product. |
| Validated Photostability Chamber | Provides controlled, uniform light output meeting ICH Q1B spectral requirements for both visible and UV regions. |
| Temperature/Humidity Data Logger | Monitors and records ancillary conditions inside the chamber during the long exposure period, as temperature is not controlled in ICH Q1B. |
| Light-Resistant Containers | Used to store dark control samples in the same thermal environment as the exposed samples, isolating the effect of light. |
1. Introduction Forced degradation studies are a critical component of pharmaceutical development, providing essential data on drug substance and product stability under conditions more severe than those used in confirmatory ICH Q1A(R2) stability studies. This guide positions forced degradation within the broader stability strategy, explicitly contrasting its objectives with those of confirmatory photostability testing as per ICH Q1B. While ICH Q1B outlines standardized light exposure conditions to confirm the robustness of marketed packaging, forced degradation employs exceedance strategies (higher stressor intensities) and scientifically justified time points to probe degradation pathways, identify potential impurities, and validate analytical methods for stability-indicating power.
2. Forced Degradation vs. ICH Q1B Confirmatory Studies: A Conceptual Framework The design of forced degradation experiments is fundamentally driven by different goals than confirmatory photostability testing. The table below delineates the core differences.
Table 1: Contrasting Objectives and Designs of Forced Degradation and ICH Q1B Studies
| Aspect | ICH Q1B Confirmatory Photostability | Forced Degradation (Photolysis) |
|---|---|---|
| Primary Goal | Confirm that usual packaging affords sufficient protection; support shelf-life. | Identify degradation products, elucidate pathways, validate analytical methods. |
| Condition Setting | Fixed, standardized exposure (1.2 million lux hrs, 200 Wh/m² UV). | Exceedance strategy: Intentional exceedance of ICH conditions to force degradation. |
| Sample Form | Final packaged product (if necessary, exposed product). | Drug substance, placebo, diluted drug product to isolate photochemical effects. |
| Endpoint | Meeting acceptance criteria (e.g., assay, degradants). | Generating ~5-20% degradation for meaningful profiling. |
| Regulatory Basis | ICH Q1B definitive guideline. | ICH Q1A(R2), Q2(R1), Q3B(R2) advising on stability and impurity assessment. |
3. Core Principles: Exceedance Strategies and Time Point Selection The cornerstone of effective forced degradation is the deliberate and controlled application of stress beyond standard conditions.
- Exceedance Strategies: The aim is not to simulate real-time storage but to accelerate degradation kinetics. For photostability, this means exposing samples to higher irradiance (W/m²) while ensuring spectral distribution matches sunlight (ICH option 1 or 2). For thermal stress, temperatures may be set 10°C above accelerated conditions (e.g., 60°C, 75°C). For hydrolysis, pH extremes (e.g., pH 1-2 and 9-13) are used. The key is to avoid secondary degradation pathways that would not be relevant under normal conditions.
- Time Point Rationale: Time points are not arbitrary. They are selected to generate a degradation profile that allows for kinetic analysis. A typical protocol includes multiple interim time points (e.g., 1, 3, 7, 14 days for thermal stress; 2, 5, 10, 24 hours for photolysis) to capture the appearance and possible disappearance of degradation products. The study continues until adequate degradation (typically 5-20%) is achieved or a plateau is observed, signaling the need for increased stress.
Table 2: Exemplary Exceedance Conditions and Time Points for Common Stressors
| Stress Type | Standard Condition (e.g., ICH) | Forced Degradation Exceedance Strategy | Recommended Interim Time Points |
|---|---|---|---|
| Photolysis | 1.2 M lux hrs, 200 Wh/m² (Q1B) | 2-5x UV energy (e.g., 500-1000 Wh/m²) or higher irradiance. | 25%, 50%, 100%, 200% of ICH UV dose. |
| Thermal (Solid) | 40°C ± 2°C / 75% RH ± 5% (Accelerated) | 60°C, 70°C, or 80°C (dry). | 1, 3, 7, 14, 28 days. |
| Hydrolysis (Solution) | Room temperature, label storage pH. | Acidic (0.1N HCl) & Basic (0.1N NaOH) at 40-70°C. | 1, 3, 6, 24, 48 hours. |
| Oxidation | Not defined by ICH. | 0.1%-3% H₂O₂ at room temperature or mild heat (40°C). | 1, 3, 6, 24 hours. |
4. Detailed Experimental Protocol: Photolytic Forced Degradation This protocol illustrates the application of an exceedance strategy for photostability.
Objective: To generate and characterize photolytic degradation products of a new drug substance (DS). Materials: Drug substance, 0.1N HCl/NaOH, 3% H₂O₂, acetonitrile (HPLC grade), water (HPLC grade), quartz or clear glass vials. Equipment: Photostability chamber (meeting ICH Option 1 or 2), HPLC-DAD/UV, HPLC-MS/MS, balance. Procedure:
- Sample Preparation: Prepare a dilute solution of DS (e.g., 100 µg/mL) in a suitable, transparent solvent (e.g., water:ACN 1:1). Also prepare solid samples thinly spread in a Petri dish.
- Exposure Setup: Place solution and solid samples in the photostability chamber. Include a dark control (wrapped in aluminum foil) for each sample type.
- Exceedance Conditions: Set the chamber to provide continuous cool white fluorescent (Option 2) and near-UV lamp (Option 1) exposure. Target a total UV energy of 600 Wh/m² (3x ICH Q1B minimum). Use an integrated radiometer to confirm dose.
- Time Point Sampling: Remove samples at defined intervals corresponding to 150, 300, 450, and 600 Wh/m² UV energy.
- Analysis: Immediately analyze samples by HPLC-UV/DAD. Compare chromatograms of stressed samples against dark controls. Monitor for new peaks and loss of parent compound.
- Characterization: Pool samples showing significant degradation for analysis by LC-MS/MS to obtain structural information on degradants.
- Forced Degradation Workflow: The logical progression of the study is depicted in the following diagram.
Diagram Title: Forced Degradation Study Logical Workflow
5. The Scientist's Toolkit: Key Research Reagent Solutions Table 3: Essential Materials for Forced Degradation Studies
| Item / Reagent | Function / Purpose |
|---|---|
| ICH-Compliant Photostability Chamber | Provides controlled, reproducible light exposure meeting ICH Option 1 (UV) and Option 2 (visible) spectral requirements for both confirmatory and exceedance studies. |
| Calibrated Radiometer & Lux Meter | Critical for quantifying and documenting light exposure dose (W/m², Wh/m², lux hours), ensuring accuracy in exceedance strategies. |
| High-Purity Stress Reagents (e.g., HCl, NaOH, H₂O₂) | Ensure introduced degradation is due to the intended stress, not impurities in the reagents. |
| Inert & Transparent Sample Vessels (Quartz, Borosilicate Glass) | For photostability, vessels must not filter relevant wavelengths. For solution stress, vessels must be chemically inert. |
| Stability-Indicating HPLC/UPLC Method | The core analytical tool capable of separating and quantifying the parent compound from all degradation products. |
| LC-MS/MS System | Used for structural elucidation of unknown degradation products generated during forced degradation. |
| Controlled Stability Chambers (Ovens, Humidity Chambers) | Provide precise thermal and humidity stress conditions for solid and solution-state studies. |
6. Data Integration and Pathway Elucidation The ultimate value of forced degradation lies in linking the generated impurities to potential chemical pathways. This knowledge informs formulation development, packaging selection, and shelf-life predictions.
Diagram Title: Primary Stressors Leading to Degradation Pathways
7. Conclusion Forced degradation experimental design, centered on scientifically justified exceedance strategies and kinetic time points, is a proactive investigative tool. It exists in a complementary, yet distinct, sphere from confirmatory ICH Q1B testing. By intentionally pushing materials beyond standard conditions, scientists can map the stability landscape of a drug molecule, ensuring that confirmatory stability studies and final packaging are built upon a foundation of comprehensive mechanistic understanding. This approach is indispensable for robust analytical method validation, formulation screening, and overall risk mitigation in pharmaceutical development.
Within the framework of ICH Q1B photostability testing, a critical distinction exists between forced degradation studies and confirmatory testing. Forced degradation, a development activity, intentionally stresses a drug substance or product to elucidate potential degradation pathways and validate analytical methods. Confirmatory testing, however, demonstrates the inherent photostability of the final packaged product under standardized light conditions. This whitepaper addresses the analytical method development and validation challenges specific to identifying and quantifying photodegradants generated during forced degradation studies. The suitability of these methods—particularly specificity—is paramount, as it directly informs the reliability of stability-indicating methods used in confirmatory studies and shelf-life determination.
Core Challenges: Specificity and Detection
Photodegradants present unique analytical hurdles:
- Structural Similarity: Photoproducts are often isomers or minor structural variants of the parent drug, making chromatographic separation (specificity) difficult.
- Low Abundance: In early forced degradation, degradants may be present at very low levels (<0.1%), demanding high analytical sensitivity.
- Transient and Unstable Intermediates: Some photodegradants may be unstable under analytical conditions, leading to underestimation.
- Lack of Reference Standards: Isolated, purified photodegradant standards are rarely available, complicating identification and quantification.
Method Suitability Parameters and Experimental Protocols
Method suitability is demonstrated through validation per ICH Q2(R1) guidelines, with emphasis on specificity.
Table 1: Key Analytical Validation Parameters for Photodegradant Methods
| Parameter | Objective for Photodegradants | Acceptance Criteria Considerations |
|---|---|---|
| Specificity | Resolve all degradants from each other and the API. No co-elution. | Baseline separation (R_s > 2.0) of critical peak pairs. Peak purity tools (DAD/MS) confirm homogeneous peaks. |
| Detection Limit (LOD) | Detect trace-level degradants. | Signal-to-Noise (S/N) ≥ 3. Often 0.05% relative to parent concentration. |
| Quantitation Limit (LOQ) | Precisely quantify degradants at reporting threshold. | S/N ≥ 10. RSD of precision at LOQ ≤ 10%. Often 0.1% relative level. |
| Linearity & Range | Accurate quantification from LOQ to above expected levels. | Range from LOQ to 5-10% of parent. Correlation coefficient r² > 0.995. |
| Accuracy/Recovery | Measure correctness of degradant quantification. | Spiked recovery at multiple levels (e.g., 0.1%, 1%, 5%) within 90-110%. |
Detailed Protocol: Forced Photodegradation and Specificity Assessment
A. Sample Preparation:
- Expose a solution of the drug substance (e.g., 1 mg/mL in appropriate solvent) and solid drug substance in a quartz dish to a calibrated light source per ICH Q1B Option 2 (e.g., 1.2 million lux hours of visible light and 200 watt hours/m² of UV).
- Include dark controls (wrapped in aluminum foil) for comparison.
- Periodically withdraw samples for analysis to monitor degradation progression.
B. Specificity Evaluation via HPLC-DAD-MS:
- Chromatographic Separation:
- Column: C18 (150 x 4.6 mm, 3.5 µm) or suitable alternative.
- Mobile Phase: Gradient elution from aqueous buffer (e.g., 0.1% formic acid) to organic modifier (acetonitrile or methanol).
- Flow Rate: 1.0 mL/min.
- Detection: Diode Array Detector (DAD) scanning 200-400 nm; Mass Spectrometer with Electrospray Ionization (ESI).
- Analysis:
- Inject stressed samples, unstressed controls, and blank solvent.
- Peak Purity Assessment: Use DAD software to compare spectra across the peak (apex, upslope, downslope). A pure peak shows a match factor > 990.
- Identification: Use MS and MS/MS data to propose structures for major degradants (>0.1%).
- Forced Degradation Specificity Challenge: Co-inject the photostressed sample with the unstressed API to confirm resolution of degradant peaks from the main peak.
Visualizing the Workflow and Relationship
Title: Photodegradant Method Suitability Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Photodegradant Analysis
| Item | Function & Rationale |
|---|---|
| ICH Q1B-Compliant Light Cabinet | Provides controlled, reproducible exposure to UV and visible light per regulatory guidelines for forced degradation. |
| Quartz Suprasil Cuvettes/Dishes | High UV transparency ensures effective photolytic stress; used for solution-state exposures. |
| High-Purity HPLC Solvents & Buffers | Minimize baseline noise and ghost peaks, crucial for detecting low-abundance degradants at LOD/LOQ. |
| Photostable HPLC Vials (Amber/Certified) | Prevents artifactual photodegradation of samples waiting in the autosampler tray. |
| Mass Spectrometry Grade Ion-Pair Reagents | If needed, reagents like TFA or HFBA must be MS-compatible for LC-MS identification work. |
| Forced Degradation Sample Set | Includes stressed samples (varying time points), unstressed control, and process blanks. The primary material for method challenge. |
| Peak Purity Software (e.g., Empower, Chromeleon) | Essential tool for analyzing DAD data to confirm a single component within a chromatographic peak. |
1. Introduction: Thesis Context
The strategic application of photostability testing within pharmaceutical development is pivotal. This case study positions the empirical data within a broader thesis on ICH Q1B testing, which argues for a paradigm shift: photostability studies must evolve from mere confirmatory exercises (verifying package suitability) to integral, proactive forced degradation research. This approach enables the systematic identification of photodegradation pathways and products early in development, informing robust formulation design and analytical control strategies for drug substances and diverse solid oral dosage forms.
2. The Confirmatory vs. Forced Degradation Framework
ICH Q1B outlines a standard confirmatory test to demonstrate that light exposure does not cause unacceptable change. However, this thesis advocates for a preceding, more intensive investigative phase.
| Aspect | Confirmatory Testing (ICH Q1B) | Forced Degradation (Proposed) |
|---|---|---|
| Primary Goal | Verify suitability of proposed packaging. | Elucidate degradation pathways, identify products, establish photosensitivity. |
| Timing | Late-stage (primary packaging defined). | Early development (pre-formulation/formulation). |
| Exposure | Minimum of 1.2 million lux hours & 200 Wh/m² UV. | Exceeds confirmatory conditions; graded exposures. |
| Sample State | Final product in immediate container/closure. | Drug substance; exposed/uncoated cores; final product. |
| Outcome | Pass/Fail for packaging. | Mechanistic understanding, validation of analytical methods, formulation optimization. |
3. Experimental Protocols & Data Presentation
3.1. Core Protocol for Drug Substance & Tablet Cores
- Apparatus: Photostability chamber controlling temperature (e.g., 25°C) and equipped with both cool white fluorescent (VIS) and near-UV (e.g., UVA 320-400 nm) lamps.
- Sample Preparation: For Drug Substance (DS): spread evenly in a shallow transparent quartz or borosilicate glass dish (≤3mm thickness). For Immediate-Release (IR) Tablet Cores: remove coating (if any) and place cores on a tray.
- Exposure: Position samples alongside a calibrated radiometer/lux meter. Expose to a minimum of ICH Q1B conditions (Option 2: 1.2 million lux hours visible & 200 Wh/m² UV). For forced degradation, include intermediate time points (e.g., 25%, 50%, 100% of total exposure).
- Control: Protect identical samples with aluminum foil or opaque cover in same chamber.
- Analysis: Assess at each time point for:
- Physical: Appearance, color (colorimetric coordinates), dissolution (for tablets).
- Chemical: Potency by HPLC/UV, related substances (HPLC/PDA, LC-MS), degradation product identification.
3.2. Protocol for Coated Tablets
- Procedure: As above, but test both intact final product and, separately, tablets where a portion of the coating is carefully removed to expose the core. This assesses the protective efficacy of the coating and opacity of excipients.
- Key Comparison: Compare degradation profile of exposed core vs. intact tablet to quantify coating's photoprotective effect.
3.3. Case Study Data Summary The following table summarizes hypothetical but representative quantitative findings from a forced degradation study on a model compound, "API-X."
Table 1: Forced Degradation Results for API-X Across Product Forms
| Sample Form | Total Exposure | % Assay Remaining | Total Degradation Products (Area%) | Key Degradant (Area%) | Color Change (ΔE*ab) | Dissolution (f2 vs. control) |
|---|---|---|---|---|---|---|
| Drug Substance | 200 Wh/m² & 1.2 Mlux-hr | 85.2% | 8.7% | Photohydrate (5.2%) | 15.6 (Significant) | N/A |
| IR Tablet Core | 200 Wh/m² & 1.2 Mlux-hr | 91.5% | 5.1% | Photohydrate (3.1%) | 8.4 (Noticeable) | 52 (Fail) |
| Coated Tablet (Intact) | 200 Wh/m² & 1.2 Mlux-hr | 99.1% | 0.9% | Not Detected | 1.2 (Minimal) | 78 (Pass) |
| Coated Tablet (Core Exposed) | 200 Wh/m² & 1.2 Mlux-hr | 90.8% | 6.3% | Photohydrate (3.8%) | 10.5 (Noticeable) | 55 (Fail) |
4. The Scientist's Toolkit: Essential Research Reagents & Materials
| Item | Function/Role in Photostability Studies |
|---|---|
| Controlled Photostability Chamber | Provides calibrated, reproducible exposure to visible and UV light per ICH Q1B specifications. |
| Calibrated Radiometer/Lux Meter | Quantifies UV irradiance (W/m²) and visible illuminance (lux) to ensure accurate dosing. |
| Quartz or Borosilicate Glass Dishes | Inert, transparent containers for drug substance that do not filter relevant UV light. |
| Opaque Control Chambers (e.g., foil wraps) | Creates light-protected controls stored under identical temperature/humidity conditions. |
| HPLC-PDA (Photodiode Array) System | Primary tool for quantifying potency loss and detecting/characterizing degradation products via UV spectra. |
| LC-MS/MS (Liquid Chromatography-Tandem Mass Spectrometry) | Critical for elucidating structures of unknown photodegradants. |
| Colorimeter | Objectively measures color changes (e.g., CIELAB ΔE*) in solid samples. |
| Dissolution Test Apparatus | Evaluates impact of photodegradation on the performance of solid dosage forms. |
5. Visualizing the Workflow and Pathways
Diagram 1: Integrated photostability testing workflow.
Diagram 2: Common photodegradation pathways & excipient roles.
6. Conclusion
This case study demonstrates that applying ICH Q1B principles as a forced degradation tool, rather than solely a confirmatory test, generates critical data. For API-X, the drug substance is moderately photosensitive, forming a photohydrate. The IR core offers marginal protection via excipient screening. The coated tablet provides excellent protection, but only when the film coat is intact—exposure of the core leads to significant degradation. This mechanistic understanding, visualized through the pathways and workflow, is essential for developing stable formulations and defining appropriate controls, ultimately validating the core thesis that proactive photostability research is indispensable in modern drug development.
Within the broader thesis on ICH Q1B photostability testing, distinguishing between forced degradation (stress) studies and confirmatory (formal) stability studies is critical. Each serves a distinct purpose in drug development and requires specific documentation and reporting standards to ensure regulatory compliance and scientific validity.
Core Study Types: Objectives and Essential Data
Confirmatory (Formal) Photostability Studies
Objective: To establish the inherent photostability characteristics of a new drug substance or product under standard ICH Q1B conditions, supporting shelf-life, labeling, and packaging claims. Regulatory Basis: ICH Q1B "Photostability Testing of New Drug Substances and Products."
Essential Data to Document:
- Sample Information: Drug substance/product name, batch number, physical form, concentration/strength, and presentation (e.g., capsule, powder, solution).
- Light Source & Calibration: Detailed description of the light source (e.g., Option 1: cool white fluorescent & near-UV; Option 2: ID65/D65). Documented radiometric/actinometric calibration data confirming exposure meets the 1.2 million lux hours and 200-watt hours/m² requirements.
- Sample Configuration: Precise arrangement of samples and controls (protected by foil wrap) within the chamber, including distance from the source.
- Chamber Conditions: Temperature and humidity monitoring data throughout the exposure period.
- Analytical Results: Comprehensive data for all tested samples (exposed, dark control, protected control) at the defined timepoints. This includes:
- Physical changes (appearance, color, clarity).
- Chemical assay (potency).
- Degradation products (impurity profile).
- Assessment & Conclusion: A clear statement on whether the product meets acceptance criteria, implications for packaging and labeling, and justification for any proposed protective measures.
Forced Degradation (Stress) Photostability Studies
Objective: To identify potential degradation products, elucidate degradation pathways, and validate the specificity of analytical methods. It is a development tool, not a regulatory submission study per ICH Q1B, but its documentation is crucial for regulatory filings (e.g., in stability summaries and analytical validation reports). Thesis Context: These studies provide the mechanistic understanding that informs the design of confirmatory studies.
Essential Data to Document:
- Stress Rationale: Scientific justification for the conditions chosen (e.g., extreme light intensity, extended duration) beyond ICH Q1B minimums.
- Stress Conditions: Exact light source, intensity (measured), exposure duration, temperature, and sample form (solid, solution, thin film).
- Degradation Kinetics: Time-course data showing the progression of degradation (loss of active, formation of impurities).
- Degradant Characterization: Data supporting the identification and structural elucidation of major photodegradants (e.g., LC-MS, NMR data).
- Methodology Validation Evidence: Data demonstrating that the analytical method can adequately separate and quantify the active ingredient from its photodegradants (peak purity, resolution).
- Pathway Proposal: A reasoned degradation pathway based on the identified products.
Summarized Comparative Data
Table 1: Core Comparison of Confirmatory vs. Forced Degradation Photostability Studies
| Feature | Confirmatory (ICH Q1B) Study | Forced Degradation Study |
|---|---|---|
| Primary Goal | Regulatory compliance; define labeling/packaging | Understand degradation pathways; validate methods |
| Regulatory Driver | ICH Q1B (Mandatory) | ICH Q2(R2), Q1A (Expected) |
| Sample Presentation | Final marketed form & minimally exposed | Various forms (solid, solution) to maximize degradation |
| Light Exposure | Strictly controlled to ICH minimums (1.2 M lux-hr, 200 Wh/m²) | Exceeds ICH minimums; variable intensities & durations |
| Acceptance Criteria | Pre-defined limits for assay, impurities, appearance | No pre-set acceptance criteria; seek significant degradation (~5-20% loss) |
| Key Output | Pass/Fail stability assessment | Degradation products, pathway, validated stability-indicating method |
Table 2: Essential Analytical Data for Reporting
| Data Category | Confirmatory Study | Forced Degradation Study |
|---|---|---|
| Potency Assay | Quantitative results vs. specification. | Percent depletion over time. |
| Impurity Profile | Levels of specified and unspecified degradants vs. qualification thresholds. | List and proposed structures of major degradants; mass balance. |
| Physical Attributes | Documented color, appearance changes (e.g., via color scale). | Descriptive notes on physical changes. |
| Supporting Evidence | Chamber calibration certificates. | Chromatograms showing peak purity, resolution; spectral data (UV, MS). |
Detailed Experimental Protocols
Protocol 1: Confirmatory Photostability Study per ICH Q1B
- Calibration: Confirm light source output using a calibrated radiometer/lux meter and actinometer (e.g., quinine chemical actinometry for UV) prior to study.
- Sample Preparation: Prepare a single batch of drug product in its final primary packaging. Also test the drug substance if applicable. Prepare two identical sets: one for light exposure and one as a dark control (wrapped in aluminum foil).
- Chamber Loading: Place samples in the photostability chamber, ensuring uniform exposure. Include protected controls. Monitor and record temperature continuously.
- Exposure: Expose samples per ICH Option 1 (simultaneous cool white & near-UV) or Option 2 (full-spectrum daylight). Continue until the integrated exposure reaches 1.2 million lux hours (visible) and 200 watt-hours/m² (UV).
- Analysis: At study completion, analyze exposed and control samples concurrently for appearance, potency, and degradation products using validated methods.
- Reporting: Compile all data, comparing results against predefined acceptance criteria. Conclude on the need for protective packaging.
Protocol 2: Photolytic Forced Degradation Study
- Condition Scouting: Expose drug substance (in solid state and in solution at various pHs) to intense light (e.g., ~3-5x ICH intensity). Sample at intervals (e.g., 24, 48, 120 hrs).
- Sample Preparation: For solutions, prepare in quartz vials to allow full UV transmission. For solids, spread thinly in open petri dishes.
- Stress Execution: Place samples in a light cabinet or solar simulator. Maintain constant temperature (e.g., 25°C). Protect a separate set of controls in the dark.
- Monitoring: Analyze samples periodically by HPLC-UV/PDA to track degradation progress. Target ~10-20% degradation of the active for optimal degradant formation.
- Degradant Identification: Isolate major degradants via preparative HPLC. Characterize using LC-MS/MS, high-resolution MS, and NMR spectroscopy.
- Method Suitability: Demonstrate that the analytical method resolves all major degradants from the main peak and from each other (resolution >1.5), and provides accurate peak purity (PDA assessment).
Visualizing the Photostability Testing Workflow
Title: Photostability Testing Strategy in Drug Development
Title: Generalized Photodegradation Pathways
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Photostability Testing
| Item | Function & Rationale |
|---|---|
| Controlled Light Chamber | Provides calibrated, uniform exposure to visible and UV light as per ICH Q1B options. Must allow for temperature control. |
| Calibrated Radiometer / Lux Meter | Measures visible light intensity (lux) to calculate cumulative exposure (lux-hours). Essential for protocol compliance. |
| Chemical Actinometer (e.g., Quinine) | A chemical solution whose photochemical yield is precisely known. Used to calibrate UV light intensity (watt-hours/m²) in the chamber. |
| HPLC System with PDA Detector | The primary analytical tool. Separates degradants from the API. The Photodiode Array (PDA) detector is critical for assessing peak purity and identifying degradants. |
| LC-MS/MS System | Used primarily in forced degradation studies to determine the molecular weight and propose fragmentation patterns of photodegradants. |
| Quartz Suprasil Cuvettes/Vials | For solution-phase forced degradation. Quartz transmits full UV spectrum, unlike standard glass, ensuring relevant photochemistry. |
| Aluminum Foil | Used to wrap protected control samples, providing a critical dark control to distinguish light-induced changes from thermal effects. |
| Validated Stability-Indicating HPLC Method | An analytical method proven to accurately measure the API while resolving it from all known and potential degradants. The cornerstone of both study types. |
Overcoming Photostability Challenges: Troubleshooting Failed Studies and Optimizing Experimental Design
Confirmatory photostability testing, as the final validation step per ICH Q1B, serves a distinct purpose from forced degradation studies. Forced degradation is a development tool used to elucidate degradation pathways and validate analytical methods. In contrast, confirmatory testing provides definitive evidence that standard lighting conditions do not adversely affect the drug product within its shelf life. The pitfalls discussed herein—inadequate coverage and sample handling errors—directly threaten the validity of this regulatory submission data, potentially leading to incorrect stability conclusions and patient risk.
The Pitfall of Inadequate Coverage
Inadequate coverage refers to the failure to expose all critical components of a drug product to the required light energy, compromising the test's comprehensiveness.
Quantitative Data: Coverage Deficiencies in Common Packaging
The following table summarizes data from recent studies on light penetration.
Table 1: Light Transmission through Common Primary Packaging Materials (Simulated ICH Option 2 Conditions)
| Packaging Material | Thickness (mm) | UV (320-400 nm) Transmission (%) | Visible (400-800 nm) Transmission (%) | Common Product Applications |
|---|---|---|---|---|
| Clear Type I Glass | 1.0 | <1% | >90% | IV Solutions, Vials |
| Amber Type I Glass | 1.0 | <0.1% | 10-25% | Light-Sensitive Solutions |
| Polypropylene (Clear) | 0.5 | ~5% | ~85% | Tablets, Capsules in Blisters |
| Polyvinyl Chloride (Clear) | 0.2 | ~15% | ~92% | IV Bags |
| Aluminum Foil | 0.03 | 0% | 0% | Blister Lidding, Sachets |
| HDPE (White Opaque) | 0.8 | 0% | <5% | Bottles for Solids |
Source: Compiled from recent pharmaceutical packaging studies (2022-2024).
Experimental Protocol: Validating Full Product Coverage
Protocol: Mapping Light Exposure in Secondary Packaging
- Objective: To confirm that all units within a secondary pack (e.g., carton) receive adequate illumination during confirmatory testing.
- Materials: Lux/UV radiometer with data logging, mock drug product units, cardboard carton, ICH-compliant light cabinet.
- Method:
- Place radiometer sensors inside representative mock units (e.g., vial, blister cavity).
- Position these units at various locations within the secondary carton: center, edges, top, and bottom.
- Load the carton into the photostability chamber per ICH Q1B guidelines (Option 1 or 2).
- Initiate exposure and continuously log irradiance data from each sensor for the duration of the test.
- Calculate cumulative exposure for each location and compare to the target (e.g., 1.2 million lux hours, 200 W·h/m² UVA).
- Critical Analysis: This protocol often reveals "shadowing" effects, where units in the center receive up to 40% less visible light than those on the periphery, invalidating the assumption of uniform coverage.
Diagram 1: Logical pathway from inadequate coverage to false negative conclusion.
The Pitfall of Sample Handling Errors
Post-exposure handling is critical. Degradation products formed during photostability testing can be transient or photosensitive, and improper handling can lead to their loss or transformation, skewing results.
Quantitative Data: Impact of Handling Variables on Analyte Recovery
Table 2: Effect of Post-Exposure Handling Conditions on Measured Degradant Levels
| Handling Variable | Condition A | Condition B | % Change in Key Degradant | Typical Impact on Potency Assay |
|---|---|---|---|---|
| Storage Temp (Before Analysis) | 4°C, protected light | 25°C, ambient light | -35% to -80% | Overestimation of stability |
| Time-to-Analysis Delay | 24 hours | 7 days | -25% to -50% | Overestimation of stability |
| Extraction Solvent (for solids) | Aqueous Buffer, N₂ Sparge | Aqueous Buffer, Air | -15% | Underestimation of oxidation |
| Container (for solutions) | Amber Glass, Headspace Minimized | Clear Glass, Normal Headspace | +200% (for some photoproducts) | Underestimation of photostability |
Source: Analysis of recent method validation and stability study discrepancies (2023-2024).
Experimental Protocol: Controlling Post-Exposure Sample Integrity
Protocol: Stabilization and Processing of Photosensitive Samples
- Objective: To ensure accurate quantification of photodegradants from exposure to analysis.
- Materials: Exposed samples, amber glass vials with PTFE-lined caps, cryogenic labels, nitrogen purge line, refrigerated centrifuge, low-actinic glassware.
- Method:
- Immediate Stabilization: Upon removal from the light cabinet, immediately transfer samples to a pre-cooled (4°C), low-light environment.
- Primary Packaging Handling: For products in immediate containers, open them under controlled, low-intensity light (e.g., red safelight for some compounds).
- Sample Preparation: For assay, rapidly dissolve or extract the sample using deoxygenated solvent (sparged with nitrogen or argon) in an amber volumetric flask.
- Intermediate Storage: If analysis cannot be immediate, freeze the prepared solutions at ≤ -20°C in amber vials with minimal headspace. Document and justify any hold time.
- Analysis: Thaw samples under controlled conditions and analyze using a stability-indicating method (e.g., HPLC with PDA). Include control samples (dark controls) processed identically.
Diagram 2: Workflow for handling photosensitive samples post-exposure.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Robust Confirmatory Photostability Testing
| Item | Function/Application | Key Consideration |
|---|---|---|
| Calibrated Radiometer (Lux & UV-A) | Measures light intensity inside chamber and at sample positions to verify ICH compliance. | Must be NIST-traceable and calibrated annually. |
| Data-Logging Radiometer Sensors | Maps spatial distribution of light within loaded chambers and packaging to identify shadow zones. | Small sensor size is critical for placement in blisters or vials. |
| Low-Actinic Laboratory Glassware (Amber/Red) | Used for all sample preparation post-exposure to prevent additional, unintended photodegradation. | Ensure compatibility with extraction solvents. |
| Nitrogen/Argon Sparging Kit | Deoxygenates extraction solvents to halt post-exposure oxidative degradation chain reactions. | Use high-purity (≥99.99%) gas. |
| Temperature-Controlled, Light-Tight Sample Chamber | Provides a standardized holding environment for samples between exposure and analysis. | Must maintain 4±1°C. |
| Validated Stability-Indicating Assay (e.g., HPLC-PDA/UPLC-MS) | Separates and quantifies drug substance from all degradation products formed during testing. | Must be validated for specificity toward known and potential degradants. |
| Spectral Power Distribution (SPD) Meter | Confirms the light source output matches the D65/ID65 standard as required by ICH Q1B. | Critical for qualifying new light sources or chambers. |
Confirmatory testing under ICH Q1B is a definitive gatekeeper for drug product stability claims. Inadequate spatial coverage of the sample set and inadvertent post-exposure sample degradation are two pervasive, technical pitfalls that can systematically bias data towards a false conclusion of stability. By implementing mapping protocols for coverage, stringent sample handling workflows, and utilizing the appropriate toolkit, researchers can generate data that truly confirms the photostability of the product in its marketed packaging, ensuring patient safety and regulatory compliance. This rigorous approach distinguishes confirmatory testing from its exploratory counterpart, forced degradation research.
1.0 Introduction and Thesis Context
Within the framework of ICH Q1B photostability testing, forced degradation (or stress testing) serves a distinct purpose from confirmatory studies. The confirmatory study, as per ICH Q1B, aims to demonstrate the inherent stability of a drug product under specified lighting conditions. In contrast, forced degradation is a development tool, intentionally applying severe stress (thermal, photolytic, oxidative, hydrolytic) to elucidate degradation pathways, identify potential impurities, and validate analytical methods. This whitepaper posits that a result of "no change" or "no degradation" in a forced degradation study is not a definitive endpoint but a critical data point requiring systematic interpretation. Its meaning is contingent upon two interdependent variables: the severity of the applied insult and the sensitivity of the analytical methods employed. Misinterpretation can lead to false confidence in product stability or analytical control strategies.
2.0 Core Concepts: Insult Severity and Analytical Sensitivity
2.1 Quantifying Insult Severity Insult severity must be measurable and justified beyond standard ICH Q1B confirmatory conditions. It is defined by the intensity and duration of the stress factor.
Table 1: Comparative Stress Conditions for Photolytic Forced Degradation
| Stress Parameter | ICH Q1B Confirmatory Study | Typical Forced Degradation "Severe" Insult | Rationale for Severity |
|---|---|---|---|
| Illuminance | 1.2 million lux hours (Visible) | 3-5 million lux hours | Exceeds standard by 3-5x to provoke degradation. |
| UV Energy | 200 watt hours/m² (UV) | 600-1000 watt hours/m² | Exceeds standard by 3-5x to challenge photolytic pathways. |
| Temperature | Ambient (per product storage) | 40-50°C (in conjunction with light) | Accelerates solid-state or solution-state reactions. |
| Sample Form | Final packaged product | Powder, thin film, or concentrated solution | Removes protective packaging and increases exposed surface area. |
2.2 Defining Analytical Sensitivity Sensitivity is the ability to detect and quantify change. "No change" is only meaningful relative to the detection threshold of the analytical suite.
Table 2: Key Analytical Method Parameters for Interpreting "No Change"
| Analytical Method | Critical Sensitivity Parameter | Typical Target for Forced Degradation | Function in Interpretation |
|---|---|---|---|
| HPLC/UHPLC-UV | Limit of Detection (LOD) / Quantification (LOQ) for degradants | LOD ≤ 0.05% of API | Primary tool for quantifying degradation products. |
| HPLC-MS/MS | Mass accuracy and MS sensitivity | Identification of degradants at 0.01% level | Structural elucidation of trace degradants. |
| Content Assay | Method precision and accuracy | Ability to detect 1-2% loss in potency | Measures main component loss. |
| Peak Purity Tools (DAD, MS) | Spectral contrast threshold | Confirms main peak homogeneity | Distinguishes co-eluting degradants from "no change". |
3.0 Experimental Protocols for Systematic Investigation
When "no change" is observed, a tiered experimental approach is required to rule out insufficient insult or inadequate sensitivity.
Protocol 1: Escalated Photostress Testing
- Objective: To determine the threshold of degradation.
- Method:
- Prepare samples as a thin film (neat API) or in a concentrated, photolabile solvent (e.g., 0.1 mg/mL in methanol/water).
- Expose samples to incrementally increased light energy (e.g., 1, 3, 5, 10 million lux hours).
- Analyze after each interval using a stability-indicating HPLC method with peak purity analysis (DAD).
- Include a validated positive control (e.g., known photolabile substance like nifedipine).
Protocol 2: Coupled Stress Testing
- Objective: To evaluate synergistic effects.
- Method:
- Expose solid API or product to high-intensity light per Protocol 1 while controlling chamber temperature at 50°C ± 2°C.
- Alternatively, subject a drug product to ICH Q1B light conditions while maintaining 75% RH.
- Analyze for changes in appearance, assay, and degradants.
Protocol 3: Analytical Orthogonality and Pre-concentration
- Objective: To enhance detection sensitivity.
- Method:
- After severe stress, concentrate the sample solution by a factor of 10-50 (e.g., via nitrogen blow-down or solid-phase extraction).
- Analyze the concentrated sample using the primary HPLC method.
- Employ an orthogonal separation (different column chemistry, e.g., HILIC vs. reversed-phase).
- Utilize LC-MS with non-targeted screening (e.g., mass defect filtering) to identify unexpected degradants.
4.0 The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Forced Degradation Studies
| Item | Function & Rationale |
|---|---|
| Controlled Light Exposure Chamber | Provides precise, ICH Q1B-compliant control of lux and watt-hour metrics for reproducible insult severity. |
| Photolytic Positive Controls (e.g., Nifedipine, Riboflavin) | Validates the stress system's ability to induce degradation, confirming the experimental setup is functioning. |
| High-Purity Solvents & Inert Atmospheres | Ensures observed degradation is due to light/heat, not solvent radicals or oxidative artifacts. |
| Stability-Indicating Method Columns | Columns resistant to high pH or varied mobile phases for method development orthogonal to primary assay. |
| LC-MS Grade Mobile Phase Additives | Volatile buffers (ammonium formate/acetate) enable seamless transition from HPLC-UV to MS for identification. |
| Forced Degradation Software Suites | Tools for kinetic modeling, predicting degradation pathways, and managing large stress study data sets. |
5.0 Decision Pathways and Data Interpretation
Diagram Title: Decision Logic for Interpreting "No Change" Results
6.0 Conclusion
In the context of ICH Q1B, a "no change" outcome in forced degradation studies is a call for rigorous investigation, not a conclusion. It necessitates a verified demonstration that the applied stress was sufficiently severe to cleave the weakest molecular bonds and that the analytical methods were sufficiently sensitive to detect the resulting changes. By systematically escalating insult severity and employing orthogonal, sensitive analytics, scientists can accurately distinguish between a truly stable molecule and a false negative. This rigorous approach ensures that forced degradation studies fulfill their essential role in robust pharmaceutical development, informing formulation, packaging, and control strategies, far beyond the confirmatory aims of standard photostability testing.
The ICH Q1B guideline, "Photostability Testing of New Drug Substances and Products," establishes a framework for evaluating the intrinsic stability of pharmaceuticals when exposed to light. Within drug development, photostability testing serves two distinct but complementary purposes: forced degradation (stress testing) and confirmatory (formal stability) studies.
- Forced Degradation Studies are intensive, investigational studies designed to elucidate the intrinsic photosensitivity of a drug substance, identify potential degradation pathways and products, and validate analytical methods for their detection. Conditions often exceed standard ICH illuminations to "force" degradation.
- Confirmatory Studies are formal, standardized tests conducted under the exact conditions specified in ICH Q1B (e.g., 1.2 million lux hours of visible light and 200 watt hours/square meter of UVA). Their goal is to provide data for recommending storage conditions and protective packaging to ensure product quality throughout its shelf life.
This guide bridges the gap between these two research phases. The knowledge gained from forced degradation studies on the compound's degradation mechanisms directly informs the design of effective protective formulations and packaging strategies, which are then validated through confirmatory studies.
Key Degradation Pathways of Photosensitive Compounds
Photosensitive degradation is initiated by the absorption of specific wavelengths of light (typically UV and visible blue light) by a chromophore within the molecule. This leads to two primary pathways:
- Direct Photolysis: The excited-state molecule itself undergoes bond cleavage or rearrangement.
- Indirect Photodegradation: The excited molecule (sensitizer) transfers energy to another molecule (e.g., oxygen, excipient), generating reactive species like singlet oxygen (
¹O₂), superoxide anion (O₂⁻), or hydroxyl radicals (OH•), which then attack the drug molecule.
A common and damaging pathway for many organic compounds (e.g., dihydropyridines, tetracyclines, phenothiazines) is Singlet Oxygen-Mediated Photo-oxidation.
Diagram 1: Key Pathways in Photosensitized Degradation
Formulation Strategies for Photoprotection
The primary goal is to prevent light from reaching the active molecule or to quench its excited state.
Table 1: Quantitative Efficacy of Common Photoprotective Excipients
| Excipient Class | Example Compounds | Common Use Concentration (%) | Proposed Mechanism of Action | Key Consideration |
|---|---|---|---|---|
| Opacifiers / Light Screens | Titanium Dioxide (TiO₂), Iron Oxide (Red, Yellow, Black) | 0.5 - 2.0% (coating) | Reflects and scatters incident light physically. | Must be uniformly distributed in coating; may affect dissolution. |
| UV Absorbers | Benzophenones (e.g., Eusolex 4360), Phenylbenzimidazole Sulfonic Acid | 0.05 - 0.5% | Absorbs UV radiation and dissipates it as heat. | Must have higher molar absorptivity than API at same λ; potential for sensitization. |
| Antioxidants / Quenchers | α-Tocopherol, Ascorbic Acid, Butylated Hydroxyanisole (BHA) | 0.01 - 0.1% | Scavenges reactive oxygen species (ROS) or quenches excited-state API. | Must be compatible; may itself degrade; requires optimization. |
| Chelating Agents | Edetate Disodium (EDTA), Citric Acid | 0.01 - 0.1% | Binds trace metal ions (Fe³⁺, Cu²⁺) that catalyze photo-oxidation. | Effective in solution or moist solid-state. |
| Cyclodextrins | β-Cyclodextrin, Hydroxypropyl-β-CD (HPβCD) | 1:1 or 2:1 Molar Ratio (CD:API) | Forms inclusion complex, shielding chromophore within hydrophobic cavity. | Can alter solubility, stability, and bioavailability; requires stoichiometric optimization. |
Experimental Protocol: Screening Photoprotective Excipients via Forced Degradation
- Objective: To rank the efficacy of various photostabilizers.
- Materials: Drug substance, candidate excipients (see Table 1), volatile solvent (e.g., methanol, acetone).
- Method:
- Prepare thin, uniform films of drug + excipient (at target ratios) on inert surfaces (e.g., watch glasses) via solvent evaporation.
- Expose samples alongside a pure drug control in a calibrated photostability chamber under forced degradation conditions (e.g., ~3-5x ICH UV dose).
- Monitor degradation periodically (e.g., at 25%, 50%, 100%, 200% of ICH UV dose) using a validated stability-indicating method (e.g., HPLC-UV/PDA).
- Calculate degradation rate constants or percent remaining relative to dark controls.
- Analysis: The excipient yielding the slowest degradation rate and simplest degradation profile (fewer impurities) is the most effective.
Packaging Strategies for Photoprotection
Packaging is the final and critical barrier. ICH Q1B requires testing directly in the proposed market packaging.
Table 2: Protective Performance of Common Pharmaceutical Packaging Materials
| Packaging Material | Typical Light Transmission (UV-Vis) | Protection Mechanism | Suitability & Notes |
|---|---|---|---|
| Clear Glass (Type I) | High (Transparent to >300 nm) | Minimal. | Requires opaque secondary packaging (carton). Not suitable alone. |
| Amber Glass (Type I) | Very Low (<450 nm) | Absorption of high-energy UV/blue light by iron oxide and other additives. | Industry standard for light-sensitive liquids/solids. Protects against UV & blue light. |
| Colored HDPE/PET | Variable (e.g., Amber, Green, Blue) | Absorption by colorants (pigments/dyes). | Amber HDPE bottles widely used for tablets/capsules. Color consistency is critical. |
| Aluminum Blister / Strip | Zero (Impervious) | Complete physical blockage of all light. | Offers the highest level of protection. Adds moisture and gas barrier. |
| PVC/PE Clear Blister | High | Minimal. | Must be used with a completely opaque carton. |
| Opaque Laminated Sachet | Zero (with aluminum layer) | Composite structure with aluminum foil layer blocks all light. | Used for unit-dose powders, effervescent tablets. |
Experimental Protocol: Confirmatory Photostability Testing per ICH Q1B
- Objective: To confirm the adequacy of the final formulation in its proposed packaging.
- Materials: Finished drug product in immediate container/closure system, with and without secondary carton (as applicable).
- Method:
- Place samples and dark controls (wrapped in aluminum foil) in a calibrated photostability chamber.
- Expose to minimum of 1.2 million lux hours of visible light and 200 watt hours/m² of UVA radiation, as per ICH Option 1 or 2.
- Control temperature to avoid thermal effects (typically 25°C ± 2°C).
- Analyze samples post-exposure alongside dark controls using validated methods.
- Acceptance Criteria: The product must meet all predetermined specifications (assay, degradation products, physical attributes) with an appropriate margin of safety.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Photostability Research
| Item / Reagent Solution | Function in Photostability Studies |
|---|---|
| Calibrated Photostability Chamber | Provides controlled, reproducible ICH Q1B-compliant exposure conditions (lux & W·h/m²). |
| Lux Meter & Radiometer (UVA) | Essential for calibrating the chamber and verifying light exposure doses. |
| Chemical Actinometers (e.g., Quinine monohydrochloride, Potassium ferrioxalate) | Used to validate the photonic dose received by samples, translating physical light measurements into chemical effect. |
| Stability-Indicating HPLC/PDA Method | Separates, identifies, and quantifies the drug and all its photodegradation products. PDA detects spectral purity. |
| Singlet Oxygen Scavenger (e.g., Sodium azide, DABCO) | Used in mechanistic forced degradation studies to confirm/rule out singlet oxygen-mediated pathways. |
| Deuterated or Degassed Solvents | Used in mechanistic studies to confirm solvent participation (H/D exchange) or oxygen-dependent pathways. |
| Standardized Packaging Simulants (e.g., clear/amber glass vials, opaque wraps) | For systematic screening of packaging protection during formulation development. |
Diagram 2: From Forced Degradation to Protective Strategies
This guide examines the specific photostability challenges of complex dosage forms through the dual lenses of ICH Q1B. While confirmatory testing provides a standardized, pass/fail check on the final packaged product, forced degradation studies are an essential, proactive research tool. For semisolids, parenterals, and combination products, forced degradation is indispensable for identifying photosensitive excipients, elucidating drug-excipient interactions under light stress, and informing protective formulation strategies. This document details the advanced methodologies required to adapt both confirmatory and forced degradation protocols to these complex systems.
Semisolid Dosage Forms: Creams, Gels, and Ointments
Semisolid matrices present heterogeneous environments where photodegradation kinetics are influenced by partition coefficients, emulsifier properties, and water activity.
Key Experimental Protocol: Photostability Mapping of a Topical Cream
- Objective: To spatially assess photodegradation within a cream matrix and correlate it with component distribution.
- Method:
- Prepare cream samples (~1 mm thick) in shallow Petri dishes.
- Expose under ICH Q1B Option 1 conditions (1.2 million lux hours visible, 200 Wh/m² UV).
- Employ a micro-sampling technique using a core boring needle to collect samples from the surface (0-0.5mm) and subsurface (0.5-1.5mm) layers separately.
- Analyze each layer via HPLC-PDA for drug substance and LC-MS for degradation products.
- Perform texture analysis and rheology pre- and post-exposure to assess physical stability.
Table 1: Representative Photodegradation Data in a Model Hydrocortisone Cream
| Formulation Layer | % Assay Remaining (UVA) | Main Degradant Identified | Viscosity Change (%) |
|---|---|---|---|
| Surface (0-0.5 mm) | 85.2 ± 3.1 | 11-Dehydrocorticosteroid | -12.5 |
| Subsurface (0.5-1.5 mm) | 98.7 ± 1.5 | Not Detected | -2.1 |
| Bulk (Homogenized) | 92.5 ± 2.4 | 11-Dehydrocorticosteroid | -8.3 |
Diagram Title: Semisolid Photostability Workflow
Parenteral Dosage Forms: Solutions, Suspensions, and Lyophilized Powders
Parenterals require stringent control of both chemical and physical changes upon light exposure, with a focus on subvisible particle formation and preservative stability.
Key Experimental Protocol: Forced Degradation of a Biologic Lyophilizate
- Objective: To predict long-term photostability and identify critical protective handling steps.
- Method:
- Reconstitute lyophilized product with appropriate diluent.
- Subject to accelerated light stress (e.g., 3x ICH UV dose) in both clear glass vials (primary container) and in quartz cuvettes (direct exposure).
- Analyze samples at intervals for:
- Primary Structure: Peptide mapping via UPLC-MS.
- Higher-Order Structure: Far-UV Circular Dichroism (CD) Spectroscopy.
- Aggregation: Size Exclusion Chromatography (SEC) and Microflow Imaging (MFI) for subvisible particles.
- Functionality: Cell-based bioassay.
Table 2: Forced Degradation Profile of a Model Monoclonal Antibody Solution
| Stress Condition | % Monomer (SEC) | Subvisible Particles ≥10µm/mL (MFI) | Bioactivity (%) |
|---|---|---|---|
| Initial (Control) | 99.5 | 5,200 | 100 |
| ICH Q1B Confirmatory | 98.8 | 6,100 | 98 |
| 3x UV Dose (Forced) | 92.1 | 45,800 | 75 |
| 5x UV Dose (Forced) | 85.7 | 112,500 | 60 |
Combination Products: Drug-Device Systems
These products require testing the drug component in its final delivery state, considering interactions with device materials (leachables, adsorption) and the impact of device operation on exposure.
Key Experimental Protocol: Photostability of a Pre-Filled Syringe (PFS)
- Objective: To evaluate the coupled stability of drug product and container system.
- Method:
- Fill PFS units (siliconized, with stainless-steel needle).
- Expose units in both upright and inverted orientations under ICH Q1B conditions.
- Analyze drug solution for potency and degradants.
- Perform extractables/leachables profiling on exposed rubber plunger and silicone oil via GC-MS.
- Assess functionality: glide force, break-loose force, and needle shield removal force.
Diagram Title: Combination Product Photostability Interactions
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item | Function in Photostability Studies |
|---|---|
| Quartz Cuvettes | Allows full-spectrum UV exposure for forced degradation studies without container interference. |
| Micro-sampling Tools (e.g., coring needles, micro-spatulas) | Enables stratified sampling of semisolid matrices to map degradation gradients. |
| Validated Chemical Actinometers (e.g., potassium ferrioxalate) | Quantifies and verifies the actual UV light dose delivered in non-standard chambers. |
| Specialized Leachables Kits | Provides sensitive, standardized methods for extracting and identifying container-derived photodegradants. |
| Forced Degradation Stress Chambers | Programmable chambers allowing precise control and exceedance of ICH light doses for predictive studies. |
| Subvisible Particle Analyzers (e.g., MFI, HIAC) | Critical for parenterals to quantify light-induced aggregation not detected by SEC. |
| LC-MS/MS with Photodiode Array (PDA) | Essential for identifying and quantifying unknown degradation products formed during forced degradation. |
This whitepaper provides an in-depth technical guide on optimizing forced degradation studies, a critical component of pharmaceutical stress testing as per ICH Q1A(R2) and Q1B. The context is framed within the broader thesis of ICH Q1B photostability testing, where forced degradation (predictive, method development) is distinct from confirmatory studies (formal, batch-specific). The primary challenge lies in designing stress conditions that generate relevant degradation products for analytical method validation and stability indication while minimizing the formation of artificial degradation artefacts not relevant to real-world storage.
The following tables summarize target conditions and quantitative outcomes for optimizing common stress tests, based on current industry practices and regulatory guidelines.
Table 1: Recommended Ranges for Forced Degradation Stress Conditions
| Stress Condition | Typical Range / Concentration | Target Degradation (5-20%) | Key Artefact Risk & Mitigation |
|---|---|---|---|
| Acidic Hydrolysis | 0.1–1.0 M HCl, 25–70°C, 1h–7d | API ~10-15% degradation | Over-degradation to non-relevant fragments; use milder acid (e.g., 0.1M) and shorter times initially. |
| Basic Hydrolysis | 0.1–0.5 M NaOH, 25–70°C, 1h–7d | API ~10-15% degradation | Racemization, ester saponification artefacts; control temperature (<40°C for base-labile compounds). |
| Oxidative | 0.1–3% H₂O₂, 25–40°C, 1h–24h | API ~5-10% degradation | Over-oxidation (e.g., sulfones from sulfoxides); use low concentration (0.1%) and cold temperature. |
| Thermal (Solid) | 70–105°C (dry oven), 1d–14d | API ~5-15% degradation | Melting, polymorphism changes, non-specific dehydration; stay below melting point. |
| Thermal (Solution) | 50–70°C, pH range, 1d–7d | API ~10-20% degradation | Hydrolysis artefacts from buffer catalysis; use minimally reactive buffers (e.g., phosphate). |
| Photolytic (ICH Q1B) | ≥1.2 million lux hours visible, ≥200 W·h/m² UV | Meeting ICH thresholds | Photo-thermal artefacts; control chamber temperature (typically 25°C ±2). |
| Humidity | 75% RH, 40°C, 1–4 weeks | API ~5-15% degradation | Hydrolysis, hydrate formation; include desiccant controls. |
Table 2: Analytical Monitoring Strategy for Degradation vs. Artefact Discrimination
| Analytical Technique | Primary Role in Optimization | Key Metrics for Artefact Identification |
|---|---|---|
| HPLC/UPLC with PDA | Primary quantification of % degradation and impurity profiling. | Peak purity index (<999 indicates co-elution), spectral homogeneity, appearance of new peaks under mild vs. harsh stress. |
| LC-MS (QTOF, Ion Trap) | Structural elucidation of degradants; pathway proposal. | Molecular weight, fragmentation pattern, comparison to known degradation pathways (e.g., +16 Da for oxidation). |
| GC-MS | For volatile degradants or after derivatization. | Identification of small molecule artefacts from excipient interactions or solvent degradation. |
| NMR (Stress Testing) | Definitive structure confirmation, especially for isomeric degradants. | Chemical shift changes, 2D correlations to distinguish real degradants from analytical artefacts. |
Experimental Protocols for Key Studies
Protocol 1: Tiered Photostability Forced Degradation (Aligned with ICH Q1B)
Objective: To generate relevant photodegradants for method development while avoiding photo-thermal artefacts.
- Sample Preparation: Prepare solutions of the drug substance in inert (e.g., methanol) and relevant (e.g., formulation buffer) solvents. Fill clear glass and clear/amber vials.
- Primary Light Exposure (Calibrated Chamber): Expose samples to a calibrated ICH light source (cool white fluorescent for visible, UV lamp). Maintain temperature at 25°C ±2°C using chamber cooling.
- Dose-Response Study: Remove aliquots at intervals (e.g., 50%, 100%, 150% of ICH minimum dose). Compare to dark controls wrapped in aluminum foil.
- Analysis: Analyze by UPLC-PDA immediately. Assess % degradation and profile changes.
- Secondary Thermal Control: Subject dark control samples to the same thermal profile recorded during light exposure to isolate pure thermal effects.
Protocol 2: Systematic Oxidative Stress Optimization
Objective: To generate primary oxidative degradants without over-stressing.
- Reagent Dilution Series: Prepare H₂O₂ solutions at 0.1%, 0.3%, 1.0%, and 3.0% (w/v) in the drug product matrix or a relevant buffer.
- Low-Temperature Incubation: Spike drug solution with each H₂O₂ concentration. Incigate at 5°C and 25°C in parallel. Sample at 1, 3, 6, and 24 hours.
- Quenching: Add a slight molar excess of methionine or catalase to quench residual H₂O₂ immediately before analysis to stop the reaction.
- Analysis: Monitor for the appearance of primary oxidation products (e.g., N-oxide, sulfoxide) via LC-MS. The condition yielding 5-10% of the primary oxidant with minimal secondary peaks is optimal.
Protocol 3: Hydrolytic Stress with Buffer Reactivity Assessment
Objective: To induce hydrolysis while minimizing buffer-catalysed artefacts.
- Buffer Selection: Prepare solutions at target pH (e.g., 2, 7, 10) using different buffers (e.g., HCl/KCl for pH 2, phosphate vs. Tris for pH 7, carbonate vs. borate for pH 10).
- Thermal Acceleration: Heat samples at 70°C and 40°C. Sample at 24, 48, and 96 hours.
- Control: Include a sample in water adjusted to the same pH with a minimal amount of acid/base as a non-buffered control.
- Analysis: Compare degradation rates and profiles across buffers. The buffer producing a profile most similar to the water control and real-time stability is preferred.
Visualizations
Diagram 1: Flow for Optimizing Forced Degradation Conditions
Diagram 2: ICH Q1B Forced Degradation vs Confirmatory Studies
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Forced Degradation Optimization
| Item / Reagent Solution | Function & Rationale |
|---|---|
| Controlled Photostability Chamber | Provides calibrated ICH light sources (UV & visible) with precise temperature and humidity control to separate photolytic from thermal effects. |
| High-Purity Stress Reagents | e.g., 30% H₂O₂ (stabilizer-free), 1M HCl/NaOH ampoules, to ensure stress originates from intended reagent, not impurities. |
| Chemically Inert Buffers | e.g., Phosphate, acetate at multiple pKa's. Allow pH control with minimal catalytic interference in hydrolysis studies. |
| Quenching Agents | L-Methionine, catalase (for H₂O₂); NaHCO₃ (for acid/base). Stop reactions instantly at timepoints for accurate snapshot of degradation. |
| UPLC/HPLC System with PDA | High-resolution separation coupled with photodiode array detection for quantifying degradation and assessing peak purity. |
| LC-MS System (QTOF preferred) | Provides accurate mass and fragmentation data for structural elucidation of unknown degradants and pathway proposal. |
| Forced Degradation Software | Specialist software (e.g., in Chromeleon, Empower) for designing, tracking, and analyzing matrixed stress studies. |
| Stable Reference Standards | Of known degradation products (if available) to confirm identity and validate analytical method response. |
1. Introduction: The Critical Role of Degradant Analysis in ICH Q1B Context
The ICH Q1B guideline on photostability testing of new drug substances and products provides a framework for evaluating light sensitivity, a critical quality attribute. This testing exists within a spectrum of stability assessments, spanning from forced degradation studies (stress testing) to confirmatory studies (on finalized packaged product). While confirmatory studies under the ICH conditions assess the suitability of the proposed packaging, forced degradation studies are predictive and investigative, designed to elucidate intrinsic stability and degradation pathways.
The primary analytical hurdle emerges in forced degradation studies, where the goal is to generate and characterize minor degradants—those present often at levels below 0.1% area. The reliable identification and accurate quantification of these species are paramount for developing stability-indicating methods, setting meaningful specifications, and understanding degradation chemistry, yet they present significant technical challenges.
2. Core Analytical Challenges & Strategic Solutions
The primary challenges in minor degradant analysis are sensitivity, selectivity, and structural elucidation.
- Challenge 1: Detection Sensitivity & Resolution. Low-abundance degradants are often obscured by the main analyte peak or noise.
- Solution: Employ High-Resolution Mass Spectrometry (HRMS) coupled with Ultra-High-Performance Liquid Chromatography (UHPLC). HRMS provides accurate mass measurements for empirical formula determination, while UHPLC offers superior peak capacity and sensitivity.
- Challenge 2: Definitive Structural Identification. An accurate mass alone is insufficient to distinguish between isomers or confirm a proposed structure.
- Solution: Integrate orthogonal spectroscopic techniques. Multi-stage tandem mass spectrometry (MSⁿ) provides fragmentation fingerprints. For isolable degradants (>0.5%), Nuclear Magnetic Resonance (NMR) spectroscopy remains the gold standard for full structural elucidation.
- Challenge 3: Reliable Quantitation at Trace Levels. Traditional UV detection lacks specificity and sensitivity for quantitation of co-eluting or trace-level degradants.
- Solution: Utilize Mass Spectrometry (MS) as the quantitative detector in a validated LC-MS/MS (Multiple Reaction Monitoring) method, offering superior selectivity and lower limits of quantification (LOQ).
3. Quantitative Data Summary: Method Capabilities
Table 1: Comparison of Analytical Techniques for Degradant Analysis
| Technique | Typical LOQ (% w/w) | Primary Role | Key Strength | Key Limitation |
|---|---|---|---|---|
| HPLC-UV/DAD | 0.05 - 0.10% | Primary forced degradation screening & purity assay. | Universal detection, robust, cost-effective. | Poor specificity for co-eluters, low sensitivity. |
| UHPLC-HRMS | 0.001 - 0.01% | Identification & semi-quantitation of unknown degradants. | Accurate mass, empirical formula, high sensitivity. | Requires expertise; quantitative precision less than LC-MS/MS. |
| LC-MS/MS (MRM) | 0.0001 - 0.001% | Targeted quantification of known degradants. | Exceptional selectivity and sensitivity for quantitation. | Method development required per analyte; not for unknowns. |
| Preparative HPLC | N/A (Isolation) | Isolation of degradants for further study. | Provides pure material for NMR, biological testing. | Scale-up required; time and resource intensive. |
4. Experimental Protocols for a Tiered Analytical Workflow
Protocol 4.1: Initial Forced Degradation & Screening (ICH Q1B-Informed)
- Stress Conditions: Expose solid drug substance and/or drug product (e.g., in a quartz cell) to an overall illumination of not less than 1.2 million lux hours and an integrated near-UV energy of not less than 200 watt-hours/square meter per ICH Q1B. Include appropriate dark controls.
- Analysis: Analyze stressed samples vs. controls via a generic UHPLC-UV/DAD method (e.g., C18 column, 30°C, gradient 5-95% organic over 15 min, 0.6 mL/min). Monitor at multiple wavelengths.
- Goal: Assess gross degradation and identify retention times of potential degradants.
Protocol 4.2: Identification of Minor Degradants via HRMS
- Instrumentation: UHPLC system coupled to a Q-TOF or Orbitrap mass spectrometer.
- Method: Use the same chromatographic conditions as Protocol 4.1, with mobile phases compatible with MS (e.g., formic acid/ammonium formate buffers). Employ positive/negative electrospray ionization (ESI) with data-dependent acquisition (DDA).
- Data Processing: Use software to perform mass defect filtering, background subtraction, and fragment ion analysis. Propose structures based on accurate mass of parent and fragments, and plausible degradation chemistry (e.g., oxidation, hydrolysis).
Protocol 4.3: Quantitative Method for Key Degradants
- Standards: Isolate or synthesize the identified degradant. Prepare a stock solution and serial dilutions for a calibration curve.
- Instrumentation: LC-MS/MS system (triple quadrupole).
- Method Development: Optimize MS parameters (collision energy, fragmentor voltage) for the degradant and the API (as internal standard). Establish at least two specific MRM transitions per analyte.
- Validation: Validate the method per ICH Q2(R1) for specificity, accuracy, precision, linearity, range, and LOQ/LOD to ensure reliable quantification at the required level (e.g., 0.05%).
5. Visualizing the Analytical Decision Pathway
Diagram Title: Analytical Workflow for Minor Degradants
6. The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Degradant Identification & Quantification
| Item / Reagent Solution | Function & Rationale |
|---|---|
| High-Purity Photostability Chamber | Provides controlled, ICH Q1B-compliant illumination (lux & UV) with temperature control for reproducible forced degradation. |
| MS-Grade Mobile Phase Additives (e.g., ammonium formate, formic acid) | Ensures compatibility with mass spectrometry, minimizing ion suppression and source contamination. |
| Stable Isotope-Labeled Internal Standards (e.g., ¹³C, ²H-labeled API) | Critical for precise LC-MS/MS quantification, correcting for matrix effects and recovery variability. |
| Derivatization Reagents (e.g., for carbonyls, peroxides) | Chemically tags specific degradant functional groups to enhance MS detectability or facilitate isolation. |
| Advanced Chromatography Columns (e.g., charged surface hybrid, HILIC) | Provides orthogonal separation mechanisms to reverse-phase LC for resolving polar degradants co-eluting with the API. |
| In-silico Degradation Prediction Software | Uses algorithms to predict plausible degradation pathways, guiding the interpretation of HRMS data and structure elucidation. |
7. Conclusion
Within the ICH Q1B paradigm, the move from confirmatory testing to predictive forced degradation research demands advanced analytical strategies. Overcoming the hurdles of minor degradant analysis requires a tiered, orthogonal approach. Initial UHPLC-UV screening must be seamlessly complemented by HRMS for identification and targeted LC-MS/MS for validation and quantification. This rigorous analytical lifecycle is fundamental to ensuring drug product quality, safety, and efficacy throughout its shelf life.
Validating Stability & Packaging: A Comparative Analysis of Forced Degradation and Confirmatory Data
Within the framework of ICH Q1B "Photostability Testing of New Drug Substances and Products," a critical distinction exists between Forced Degradation Studies and Confirmatory Studies. This guide provides a detailed, technical comparison of these two pivotal approaches, tabulating their objectives, experimental protocols, and analytical outputs. The content is framed within the broader thesis that these studies serve fundamentally different but complementary purposes in pharmaceutical development: one (forced degradation) is an investigative stress study to elucidate intrinsic stability and degradation pathways, while the other (confirmatory) is a formalized, controlled test to validate the robustness of marketed packaging under specific light conditions.
Table 1: Primary Goals and Regulatory Context
| Aspect | Forced Degradation (Stress Testing) | Confirmatory Study (ICH Q1B) |
|---|---|---|
| Primary Objective | To identify potential degradation products, elucidate degradation pathways, and establish the intrinsic photosensitivity of the drug substance. | To provide evidence of product quality within the shelf-life under recommended storage conditions, including specific light conditions. |
| Regulatory Driver | ICH Q1A (R2) Stability Testing, ICH Q1B (informative). Part of development. | ICH Q1B (mandatory). Part of formal stability protocol for marketing authorization. |
| Development Stage | Early development (pre-clinical/Phase I). | Late development (Phase III) and post-approval. |
| Thesis Context | Exploratory research to understand molecule behavior under extreme stress. | Control experiment to confirm the suitability of the proposed packaging. |
Table 2: Experimental Conditions and Protocols
| Parameter | Forced Degradation | Confirmatory Study |
|---|---|---|
| Light Source | Broader range; may include multiple sources (e.g., UV at 254 nm, cool white fluorescent, xenon) to probe different photoreactions. | Strictly defined per ICH Q1B: Option 1: 1.2 million lux hours of visible light and 200 watt hours/m² of UV (near 320-400 nm). Option 2: Exposure to a validated cool white fluorescent and UV lamp. |
| Sample Presentation | Often pure drug substance in solution/solid state, possibly without primary packaging. May use multiple concentrations/pH values. | Final drug product in its immediate primary packaging (e.g., vial, blister, bottle). If sensitive, testing proceeds to fully unpackaged product. |
| Duration & Intensity | Not fixed; stress is applied until significant degradation (typically 5-20% loss of active) is achieved to generate degradants for analysis. Intensity may be exaggerated. | Fixed based on achieving the specified total illuminance/UV energy. Exposure is not continued beyond this point regardless of degradation observed. |
| Controls | Dark controls (wrapped in aluminum foil) are mandatory to differentiate thermal from photolytic effects. | Concurrent dark controls are mandatory. |
| Key Protocol Steps | 1. Prepare solutions/solid samples of drug substance. 2. Expose to high-intensity light source(s). 3. Sample at intervals (e.g., 24h, 48h, 1 week). 4. Analyze for potency and degradants. | 1. Place packaged product in photostability chamber. 2. Expose to ICH-specified light conditions. 3. Monitor chamber conditions (temp, humidity). 4. Remove after specified energy dose is achieved. 5. Analyze for appearance, potency, and degradants. |
Table 3: Analytical Outputs and Data Interpretation
| Output | Forced Degradation | Confirmatory Study |
|---|---|---|
| Primary Quantitative Data | Rate of degradation (pseudo first-order rate constant, k). Extent of degradation (% loss of API). Degradant formation kinetics. | Change in assay value (% of label claim). Change in specified degradation products (against qualified limits). |
| Primary Qualitative Data | Structural identification of major degradation products (>0.1% or identification threshold). Degradation pathway elucidation (e.g., oxidation, cleavage, cyclization). | Documented changes in physical attributes (color, clarity, precipitation). Profile of degradation products (compared to reference spectra). |
| Acceptance Criteria | No pre-set acceptance limits. Goal is to generate sufficient degradation for characterization. | Must meet predefined product specification limits for assay and impurities post-exposure. |
| Impact & Decision | Informs formulation development, packaging selection, and establishes stability-indicating methods. May guide molecular redesign. | Determines if the proposed packaging is adequate. Failure necessitates packaging redesign or label storage statement changes. |
Detailed Experimental Protocols
Protocol 1: Forced Photodegradation of Drug Substance
Objective: To generate and identify major photodegradants of a new chemical entity.
- Sample Preparation:
- Prepare a 1 mg/mL solution of the drug substance in relevant solvents (e.g., water, methanol, pH 7.4 phosphate buffer).
- Aliquot 5 mL into clear 10 mL Type I glass vials.
- Prepare identical "dark control" vials wrapped completely in aluminum foil.
- Exposure Conditions:
- Place samples in a photostability chamber equipped with a cool white fluorescent lamp (emitting ~1.2 million lux) and a UV lamp (emitting ~200 W/m²).
- Maintain chamber temperature at 25°C ± 2°C.
- Expose samples for a total of 1-7 days, removing sacrificial vials at 24h, 48h, and 168h.
- Analysis:
- Potency: Analyze by HPLC-UV, comparing exposed samples to dark controls and zero-time sample.
- Degradant Profiling: Use HPLC with diode-array detector (DAD) and mass spectrometry (LC-MS/MS) for peak collection and structural identification.
- Data Calculation:
% Degradation = [1 - (Peak Area Exposed/Dark Control)] * 100.
Protocol 2: Confirmatory Photostability per ICH Q1B
Objective: To confirm the light protection offered by the marketed primary packaging.
- Sample Preparation:
- Select three batches of the final drug product.
- Place samples in their immediate transparent primary packaging (e.g., blister strips, HDPE bottles).
- Prepare dark controls in identical packaging, wrapped in foil.
- Exposure Conditions:
- Use a calibrated photostability chamber conforming to ICH Q1B Option 1 or 2.
- For Option 1: Expose until the integrated UV energy reaches 200 W·hr/m² and visible illuminance reaches 1.2 million lux·hours.
- Monitor temperature (controlled to 25°C ± 2°C) and relative humidity (ambient or controlled).
- Post-Exposure Analysis:
- Evaluate appearance (color, opacity against a white/black background).
- Perform assay (HPLC) on pooled sample units against unexposed and dark controls.
- Perform related substances analysis, specifically monitoring any new peaks or growth in known impurities.
- Acceptance:
- Product meets all registered specifications for assay and impurities.
- No significant change in appearance.
Visualizing the Photostability Testing Workflow
Title: Photostability Testing Decision Flow in Drug Development
Title: Common Photodegradation Pathway Example
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item / Reagent | Function in Photostability Testing |
|---|---|
| ICH-Compliant Light Cabinets (e.g., with cool white & UV fluorescent lamps) | Provides controlled, reproducible exposure to the specific light spectra mandated by ICH Q1B for confirmatory studies. |
| Calibrated Lux Meters & UV Radiometers | Essential for validating and monitoring the light intensity (lux) and UV energy (W/m²) inside the chamber to ensure compliance with ICH exposure limits. |
| HPLC-MS/MS System with Photodiode Array (PDA) | The core analytical instrument for separating, quantifying, and structurally characterizing the drug substance and its photodegradation products. |
| Forced Degradation Toolkit: Methanol, Acetonitrile (HPLC grade), pH Buffer Solutions (e.g., pH 3, 7, 10) | Used to prepare drug substance solutions under various conditions to probe pH-specific photodegradation pathways during forced degradation studies. |
| Validated Stability-Indicating HPLC/UPLC Method | A chromatographic method capable of resolving the API from all potential degradation products, essential for accurate potency and impurity measurement in both study types. |
| Type I Clear Glass Vials & Aluminum Foil | Standard containers for forced degradation solution studies. Foil is critical for preparing simultaneous dark controls to isolate photolytic effects from thermal effects. |
| Chemical Reference Standards (API and known impurities) | Required for method validation, peak identification, and quantifying the amount of specific degradants formed during testing. |
| Controlled Temperature/Humidity Chamber | Used for maintaining standard storage conditions (e.g., 25°C/60% RH) during long-term stability studies that often run in parallel with photostability assessments. |
Using Forced Degradation Data to Justify Confirmatory Study Acceptance Criteria
Within the ICH Q1B framework, photostability testing is a critical component of drug substance and product stability assessment. The broader thesis posits that forced degradation studies are not merely a regulatory checkbox but a foundational scientific tool. They provide the predictive data necessary to set rational, risk-based acceptance criteria for the formal confirmatory studies that follow. This guide details the methodology for leveraging forced degradation data to scientifically justify the limits set in confirmatory photostability testing.
Core Conceptual Framework
Forced degradation, under conditions more severe than those in confirmatory studies, deliberately induces photodegradation. The resulting data map the product's susceptibility, identify degradation pathways, and reveal the analytical procedure's capability. This map is then used to define appropriate "worst-case" margins for the confirmatory study's acceptance criteria, ensuring they are neither too lenient (risking patient safety) nor too stringent (leading to unnecessary specification failures).
Diagram Title: From Forced Degradation to Confirmatory Criteria
Quantitative Data Analysis & Translation to Criteria
The cornerstone of the justification lies in quantitative analysis of forced degradation results. Key parameters must be summarized and compared to proposed confirmatory limits.
Table 1: Key Forced Degradation Quantitative Metrics
| Metric | Description | Role in Justifying Confirmatory Criteria |
|---|---|---|
| Extent of Degradation (%) | Main peak loss or total degradation products formed at stress end-point. | Sets a maximum expected degradation level under severe stress; confirmatory criteria should be a fraction of this. |
| Degradation Rate | Apparent rate of degradation under high-intensity light. | Informs kinetic model; supports duration and intensity of confirmatory study. |
| Critical Degradant Identity & Level | Structure and amount of major degradation products (>0.1%). | Justifies which degradants must be specified and monitored in confirmatory studies. |
| Mass Balance | Assay value + sum of degradants vs. initial assay. | Validates analytical method; low mass balance suggests hidden degradation, demanding wider confirmatory safety margins. |
| Method Capability (S/N, LOQ) | Signal-to-Noise for degradants, Limit of Quantification. | Justifies the lower limit for reporting thresholds in confirmatory studies (e.g., reporting threshold ≥ LOQ). |
Table 2: Example Data Translation to Acceptance Criteria
| Forced Degradation Result (API, 12M J/m² UVA) | Analysis | Proposed Confirmatory Criteria (6M J/m² UVA) Justification |
|---|---|---|
| Main Potency Loss: 15% | Demonstrates intrinsic photosensitivity. | Acceptance: NMT 5% change. Justification: FD shows 15% loss under 2x confirmatory dose. Applying a 3x safety factor yields a justified limit. |
| Major Degradant "X" formed at 1.2% | Identifies critical degradant. | Acceptance: Degradant X NMT 0.5%. Justification: FD level (1.2%) provides a known worst-case. A >50% reduction from FD level is justified for formal conditions. |
| Mass Balance: 98.5% | Good recovery indicates method is stability-indicating. | Supports the use of the same method for confirmatory study without additional margin for uncertainty. |
| LOQ for Degradant X: 0.05% | Defines method sensitivity. | Justifies a reporting threshold of 0.05% and identification threshold of 0.10% for confirmatory study. |
Detailed Experimental Protocols
ICH Q1B-Aligned Forced Degradation Protocol for Justification
- Objective: To generate exaggerated degradation data for justifying confirmatory study acceptance criteria.
- Materials: Drug substance/product, appropriate transparent/opaque containers, calibrated light sources (UV-A, visible), controlled temperature chamber.
- Light Exposure: Expose samples to a total illumination of not less than 12 million lux hours (visible) and 200 watt-hours/m² (UV-A) – typically 2-3x the ICH Q1B confirmatory condition. Sample positioning per ICH Q1B options (e.g., Option 2).
- Time Points: Multiple intervals (e.g., 0, 3, 6, 12 M Lux hrs / 50, 100, 200 W-hr/m² UV) to establish kinetics.
- Control: Dark control (wrapped in aluminum foil) under same thermal conditions.
- Analysis: Assay (e.g., HPLC-UV) and related substances method at each interval. Peak purity assessment (e.g., PDA) on main peak. Stress samples vs. controls.
- Data Processing: Quantify main peak loss and all degradants > reporting threshold (e.g., >0.05%). Calculate mass balance. Plot degradation vs. light exposure.
Confirmatory Study Protocol with Justified Criteria
- Objective: To formally verify photostability under standardized ICH conditions, using acceptance criteria derived from forced degradation.
- Materials: As per forced degradation, but using final market presentation packaging.
- Light Exposure: Standard ICH Q1B confirmatory conditions: 1.2 million lux hours visible & 200 watt-hours/m² UV-A.
- Testing Points: Initial (T0) and final (after full light exposure).
- Acceptance Criteria Application: Based on Table 2 justification.
- Primary: Assay change NMT X% (justified value).
- Secondary: Specified degradant(s) NMT Y%; unspecified degradants NMT Z%; total degradants NMT T%.
- Reporting: Document results against pre-defined, justified criteria. Any OOS result is investigated against the forced degradation profile.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Photostability Testing
| Item | Function & Importance |
|---|---|
| Calibrated Light Sources | UV-A (e.g., 320-400 nm, max 365 nm) and Cool White/ID65 fluorescent lamps. Must meet ICH spectral power distribution requirements. Calibration ensures reproducible, justified stress. |
| Radiometers & Lux Meters | To measure and confirm cumulative exposure (W-hr/m² for UV, Lux-hr for visible). Critical for quantifying stress dose for correlation. |
| Controlled Temperature Chamber | Maintains samples at constant temperature (e.g., 25°C) during light exposure to isolate photolytic effects from thermal effects. |
| Validated Stability-Indicating HPLC/PDA Method | Method capable of resolving and quantifying main component and all potential degradants. PDA detector essential for peak purity and identification. |
| Photostable Reference Standard | A chemically similar compound with known photostability, used to verify light chamber performance. |
| Appropriate Sample Containers | Clear glass/plastic for forced degradation; final market packaging (clear, opaque, etc.) for confirmatory studies. Container choice is a key variable. |
Diagram Title: Justification & Study Execution Workflow
Linking Degradation Pathways to Real-World Stability Predictions
The ICH Q1B guideline for photostability testing provides a structured framework for evaluating drug substance and product stability under light exposure. A critical tension exists between the guideline's primary use for confirmatory studies—final verification of a chosen package's protective qualities—and its application in forced degradation studies—an exploratory tool to elucidate intrinsic photosensitivity and degradation pathways. This whitepaper posits that a mechanistic understanding forged in forced degradation research is the indispensable link to generating accurate, predictive models of real-world stability. By deliberately linking specific photolytic degradation pathways (e.g., oxidation, cleavage, polymerization) to quantifiable kinetic models and structural alerts, scientists can transcend the binary "pass/fail" outcome of confirmatory testing and build robust, scientifically justified stability predictions for novel formulations and storage conditions.
Core Degradation Pathways and Analytical Linkages
Forced degradation under ICH Q1B conditions (e.g., 1.2 million lux hours of visible light and 200 watt-hours/m² of UV) accelerates pathways critical for long-term stability.
Table 1: Primary Photodegradation Pathways, Analytical Signatures, and Predictive Links
| Degradation Pathway | Chemical Mechanism | Key Analytical Detection Methods | Link to Real-World Prediction |
|---|---|---|---|
| N-O Bond Cleavage | Homolytic or heterolytic cleavage of nitroso, nitrite, or N-oxide moieties. | HPLC-MS/MS (loss of NO, -16 Da fragment); LC-NMR; Increased degradants. | Predicts instability in nitro-containing APIs; Models oxygen-scavenger requirement in primary packaging. |
| Photo-oxidation | Singlet oxygen (¹O₂) or radical-mediated oxidation of alkenes, sulfides, aromatics. |
UPLC-PDA/HRMS (new peaks with +16, +32 Da); EPR for radicals; Quantification of peroxides. | Correlates to oxidative stress in formulation; Predicts need for antioxidants (e.g., BHT, ascorbate) and opaque packaging. |
| Polymerization / Dimerization | Radical recombination of photo-generated reactive species. | SEC/GPC (increased high MW species); HPLC (decreased monomer); MS for dimer mass. | Forecasts viscosity increase or particulate formation in solutions/semisolid products over time. |
| Decarboxylation | Loss of CO₂ from carboxylic acids via photo-induced electron transfer. | GC/MS for CO₂ evolution; HPLC (mass shift -44 Da); FTIR loss of C=O stretch. | Predicts pH drift and loss of potency in buffered solutions; Informs buffer system selection. |
| Ring Opening / Rearrangement | Photolysis of heterocycles (e.g., aziridines, benzodiazepines). | NMR (¹H, ¹³C) for structural change; LC-MS for isomer identification. |
Models formation of toxic or inactive isomers; Critical for potency prediction. |
Experimental Protocols: From Forced Degradation to Predictive Modeling
Protocol 1: Enhanced Photostability Forced Degradation Study
- Objective: To generate sufficient degradants for pathway identification and kinetic profiling.
- Materials: Drug substance, relevant placebo formulation, clear glass vials, ICH-compliant light cabinet (e.g., Suntest CPS+, photostability chamber).
- Procedure:
- Prepare samples in triplicate: API powder, API in solution (multiple pHs), and finished product.
- Expose samples to controlled, incremental light doses (e.g., 25%, 50%, 100%, 200% of ICH Q1B minimum) alongside dark controls.
- Maintain precise temperature (e.g., 25°C ± 2°C) and humidity control if applicable.
- At each interval, assay for potency loss and degradant formation using stability-indicating HPLC-UV/PDA.
- Isulate key degradants (>0.1%) via preparative HPLC for structural elucidation using HRMS and 2D-NMR.
- Plot degradation kinetics (log % remaining vs. light dose) to determine apparent photodegradation rate constants.
Protocol 2: Pathway-Specific Probe Studies
- Objective: To confirm reactive species and quantify their role.
- Materials: Singlet oxygen scavenger (e.g., sodium azide), radical scavenger (e.g., butylated hydroxytoluene), heavy water (D₂O, enhances
¹O₂lifetime). - Procedure:
- Subject API solution to standard light exposure in the presence and absence of specific scavengers.
- Use D₂O as solvent to probe for singlet oxygen-mediated pathways (enhanced degradation in D₂O is indicative).
- Monitor the inhibition of specific degradant formation via HPLC-MS.
- This data directly links a degradation pathway (e.g., photo-oxidation) to a mitigation strategy, enabling predictive modeling of formulation efficacy.
Protocol 3: Correlation to Real-Time Stability Conditions
- Objective: To validate the predictive model.
- Procedure:
- Using kinetic data from Protocol 1, build an Arrhenius-type model linking light energy dose to degradation rate.
- Calculate the equivalent "light stress" of proposed storage conditions (e.g., pharmacy shelf, patient in-use) based on lux-hour estimates.
- Predict the extent of degradation over the product's shelf life.
- Initiate a controlled, long-term real-time stability study under the predicted conditions.
- Periodically test samples and compare the actual degradation profile to the model prediction, refining the model coefficients.
Visualization: Workflow and Pathway Logic
Title: Linking Forced Degradation to Predictive Stability Workflow
Title: Photodegradation Pathways to Stability Prediction Logic
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Reagent Solutions for Mechanistic Photostability Research
| Item / Reagent | Function in Experiment | Rationale for Use |
|---|---|---|
| ICH-Compliant Light Cabinet | Provides controlled, reproducible exposure to visible & UV light per ICH Q1B specifications. | Standardizes the stressor for comparable, guideline-aligned forced degradation. |
| Chemical Actinometer (e.g., Quinine monohydrochloride) | Quantifies the actual photonic dose (J/cm²) received by samples. | Converts exposure time to a radiometric unit, enabling kinetic modeling across different light sources. |
| Stability-Indicating HPLC Method | Separates and quantifies API from all major degradants. | Essential for accurate measurement of degradation kinetics and impurity profiling. |
| LC-HRMS / MS-MS System | Provides accurate mass and fragmentation patterns for degradant structural elucidation. | Enables identification of degradation pathways via mass shifts and fragment ions. |
| Preparative HPLC System | Isolates individual degradants in milligram quantities. | Provides pure material for definitive structural confirmation via NMR. |
| Reactive Oxygen Species (ROS) Probes (e.g., Singlet Oxygen Sensor Green, Azide) | Identifies specific reactive intermediates (e.g., ¹O₂, radicals) involved. |
Mechanistically links physical stress (light) to chemical pathway, informing mitigation. |
| Controlled Humidity Chambers | Maintains precise %RH during light exposure for solid samples. | Evaluates critical photo-humidity interactions, a common real-world stress factor. |
| Specialized Packaging Mock-ups (e.g., UV-cut vials, amber glass, multi-layer blister) | Used in confirmatory studies informed by forced degradation results. | Directly tests the predictive model's packaging recommendation. |
Informing Primary and Secondary Packaging Decisions Based on Combined Data
Abstract This technical guide examines the integration of forced degradation and confirmatory photostability studies, framed within ICH Q1B principles, to generate robust datasets for primary and secondary packaging decisions. By combining accelerated stress data with real-time stability insights, a scientifically defensible packaging strategy that ensures drug product quality throughout its shelf life can be implemented.
1. Introduction: The ICH Q1B Framework ICH Q1B provides the foundational guideline for photostability testing of new drug substances and products. A critical distinction exists within its framework: confirmatory studies (ICH Q1B Options 1 & 2) verify the suitability of a proposed packaging under standard storage conditions, while forced degradation studies (stress testing) elucidate intrinsic photosensitivity and degradation pathways. This guide posits that only by statistically correlating data from both approaches can packaging decisions be truly informed, minimizing risk and optimizing protection.
2. Data Generation: Protocols and Integration
2.1 Forced Degradation Study Protocol Objective: To identify degradation products, establish degradation pathways, and determine the inherent photosensitivity of the drug substance and formulated product. Methodology:
- Sample Preparation: Expose drug substance (powder), placebo, and drug product (in clear glass/quartz) to controlled stress conditions.
- Light Source: A cool white fluorescent lamp (mimicking indoor light) and a near-UV fluorescent lamp (320-400 nm, λmax at 365 nm) as per ICH Q1B. Total illumination not less than 1.2 million lux hours and 200 watt hours/m² of UV energy.
- Conditions: Samples are placed in a photostability chamber with controlled temperature (e.g., 25°C). Exposure is typically continuous.
- Analysis Intervals: Samples analyzed at 0, 1.2 M Lux-hr (visible) and 200 Whr/m² (UV), and at an intermediate point. Analyses include:
- Potency assay by HPLC/UV.
- Related substances (degradant profiling) by HPLC with PDA or MS detection.
- Physical observations (color, appearance).
2.2 Confirmatory Photostability Testing Protocol Objective: To verify that the product, in its proposed market packaging, remains within specification under light conditions representative of storage and use. Methodology (Option 1 - Full Product):
- Sample Configuration: Final packaged product (in immediate container/closure system and secondary packaging, if any) is exposed.
- Light Source & Conditions: Identical to forced degradation. One set of samples is exposed to the full ICH Q1B light condition, while a second set is wrapped in aluminum foil as a dark control.
- Analysis: At the end of the exposure period, samples are compared against dark controls for potency, degradants, and physical attributes.
2.3 Data Correlation Workflow The core methodology involves the parallel execution of forced degradation and confirmatory studies, with data streams combined for a holistic analysis.
Diagram 1: Integrated Packaging Decision Workflow (76 chars)
3. Quantitative Data Summary & Decision Matrix The following tables synthesize key outputs from the combined studies to guide decision-making.
Table 1: Comparative Analysis of Forced vs. Confirmatory Study Results
| Parameter | Forced Degradation (Unpackaged) | Confirmatory Study (Packaged) | Acceptable Threshold (Example) | Decision Implication |
|---|---|---|---|---|
| Potency Loss (%) | High (e.g., >15%) | Low (e.g., <2%) | ≤5% | Packaging provides adequate protection. |
| Critical Degradant Increase | Significant formation observed. | No new or increased degradants. | Not more than (NMT) identification threshold. | Primary container barrier is sufficient. |
| Color Change | Pronounced change. | No significant change vs. dark control. | Meets product specification. | Secondary packaging may not be needed for light protection. |
Table 2: Packaging Decision Matrix Based on Combined Data
| Scenario | Forced Degradation Result | Confirmatory Result | Recommended Packaging Action |
|---|---|---|---|
| 1. High Risk | High potency loss, multiple degradants. | Significant change even when packaged. | Use opaque primary container (amber glass, opaque plastic). Mandatory protective secondary carton. |
| 2. Moderate Risk | Moderate photosensitivity. | Changes within acceptable limits in primary pack alone. | Clear or lightly tinted primary container acceptable. Secondary carton required for shelf storage. |
| 3. Low Risk | Low to no photosensitivity. | No change in clear primary pack. | Transparent primary container acceptable. Secondary packaging for physical, not light, protection. |
4. The Scientist's Toolkit: Essential Research Reagent Solutions
| Item/Reagent | Function in Photostability Studies |
|---|---|
| ICH Q1B-Compliant Light Cabinet | Provides controlled, reproducible exposure to specified visible and UV light sources. |
| Lux & UV Radiometer | Calibrated device to measure and confirm total light exposure (Lux-hr & Watt-hr/m²). |
| HPLC System with PDA Detector | Primary tool for quantifying potency loss and identifying/characterizing photodegradants. |
| Chemical Actinometers (e.g., Quinine Monohydrochloride) | Validates the photolytic output of the chamber, ensuring consistent, calibrated light energy. |
| Validated Stability-Indicating Method | HPLC or LC-MS method capable of separating and quantifying all potential degradants. |
| Appropriate Primary Container Simulants (clear glass/quartz vials) | Used in forced degradation to represent the "worst-case" exposure for the drug product. |
5. Advanced Considerations: Pathway Elucidation For products showing sensitivity, elucidating the photodegradation mechanism is critical. This often involves isolating degradants for structural elucidation (NMR, HR-MS) and mapping pathways.
Diagram 2: Photodegradation Pathway to Packaging Feedback (70 chars)
6. Conclusion Informed primary and secondary packaging decisions are not based on a single data point but on the convergence of evidence from forced degradation and confirmatory stability studies. By rigorously executing and correlating these studies within the ICH Q1B framework, drug developers can implement packaging that is both scientifically justified and commercially optimal, ensuring patient safety and product efficacy.
Within the framework of ICH Q1B "Photostability Testing of New Drug Substances and Products," a critical regulatory distinction exists between forced degradation studies and confirmatory (photostability) studies. The broader thesis posits that while confirmatory studies provide standardized, pass/fail data on the final product under specified conditions, forced degradation studies are an essential, complementary research tool. They elucidate degradation pathways, identify degradation products, and validate analytical methods. From a regulatory review perspective, assessors evaluate these complementary data sets not in isolation, but as an integrated body of evidence. This guide details how assessors scrutinize the linkage, consistency, and scientific rigor between these sets to ensure comprehensive product understanding and robust quality control.
Core Principles of Assessment: Linking Forced Degradation and Confirmatory Data
Regulatory assessors evaluate complementary data sets through several key lenses:
- Completeness and Linkage: Does the forced degradation study logically inform the conditions and analytical procedures used in the confirmatory ICH Q1B study? Assessors map degradation products found in forced studies to any products detected in confirmatory studies.
- Method Validation: Are the analytical methods used for confirmatory testing demonstrated to be "stability-indicating"? This is proven by forced degradation data showing separation of degradants from the parent compound.
- Risk Assessment & Justification: Do the data justify the light exposure conditions and product protection (e.g., packaging) chosen? Extreme forced conditions help define the margin of safety.
- Quantitative and Qualitative Consistency: Are findings across studies logically consistent? Significant degradation under forced conditions with none under confirmatory conditions supports product robustness. Inconsistencies require scientific explanation.
Table 1: Comparative Analysis of Forced Degradation vs. Confirmatory (ICH Q1B) Study Outputs
| Assessment Parameter | Forced Degradation Study (Research Tool) | Confirmatory Study (ICH Q1B Standard Test) | Regulatory Review Question |
|---|---|---|---|
| Primary Objective | Identify degradation pathways and products; validate analytical methods. | Confirm that standard lighting does not cause unacceptable change. | Does forced degradation adequately de-risk the confirmatory study? |
| Conditions | Extreme, exaggerated stress (e.g., high UV/Vis intensity, extended time). | Standardized conditions (Option 1: 1.2 million lux hrs visible, 200 W·hr/m² UV). | Are the forced conditions scientifically justified and sufficiently severe? |
| Sample Form | Drug substance (API), simple solutions/suspensions, possible product. | Final drug product in primary packaging, and API if necessary. | Are both substance and product data available and coherent? |
| Key Metrics | Degradation rate, structural identification of major degradants (>0.1%), mass balance. | Acceptance criteria change (e.g., appearance, assay, degradants). | Is mass balance from forced studies addressed? Are confirmatory specs appropriate? |
| Typical "Positive" Outcome | Significant, measurable degradation to probe pathways. | No significant change within acceptance criteria. | Does the lack of change in confirmatory testing, despite forced degradation findings, demonstrate product robustness? |
| Analytical Focus | Profiling (HPLC/UPLC-MS, NMR), method development for separation. | Validated, stability-indicating assay for quantification and impurity monitoring. | Is the confirmatory method proven to detect relevant photodegradants? |
Experimental Protocols for Key Cited Experiments
Protocol A: Forced Photodegradation for Pathway Elucidation
- Sample Preparation: Prepare separate solutions/suspensions of the drug substance (~1 mg/mL) in relevant solvents (e.g., water, pH buffers, organic/water mixes) and expose solid-state API.
- Stress Conditions: Expose samples in a calibrated photostability chamber to intense light (e.g., >5x ICH UV energy). Include dark controls. Sample at multiple time points (e.g., 24, 48, 120 hrs).
- Analysis: Analyze samples by HPLC-DAD and LC-MS/MS. Use high-resolution MS for structural identification of degradants. Calculate mass balance (assay + impurities).
- Goal: Generate a degradation profile, identify major degradation products (>0.1%), and propose a degradation pathway.
Protocol B: Confirmatory ICH Q1B Photostability Testing
- Sample Preparation: Select at least one primary batch of drug product in its immediate container/closure. Prepare similarly packaged dark controls.
- ICH Conditions: Expose samples per ICH Q1B Option 2 (worst case): total illumination of not less than 1.2 million lux hours and an integrated near ultraviolet energy of not less than 200 watt hours per square meter.
- Analysis: Analyze exposed and control samples using the validated commercial product release/stability assay. Assess against predefined acceptance criteria for appearance, assay, and degradation products.
- Goal: Demonstrate the product remains within specification when exposed to standard light conditions.
Protocol C: Cross-Validation of Analytical Methods
- Sample Set: Analyze samples from Protocol A (forced degradation) using the validated method from Protocol B.
- Evaluation: Confirm that the method resolves all major forced degradation products from the main peak and from each other (Resolution > 2.0). Verify detection and quantification limits for key degradants.
- Goal: Prove the confirmatory method is "stability-indicating" for photodegradants relevant to the product.
Visualization of Assessment Logic and Workflow
Diagram 1: Regulatory Assessment of Complementary Photostability Data
Diagram 2: Experimental Workflow for Generating Complementary Data
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Photostability Studies
| Item / Reagent | Function / Purpose | Key Considerations for Assessment |
|---|---|---|
| Calibrated Photostability Chamber | Provides controlled ICH-compliant light exposure (UV & Visible). | Review calibration certificates for lux and UV radiometer traceability. |
| ICH-Quality Light Sources | Cool White Fluorescent and Near-UV (UVA) Lamps. | Verify spectral power distribution meets ICH Q1B guidelines. |
| HPLC/UPLC with Diode Array Detector (DAD) | Primary tool for separation and quantification of degradants. | Assess method parameters (column, gradient) for resolution of degradants. |
| Liquid Chromatography-Mass Spectrometry (LC-MS/MS) | Critical for structural identification of unknown degradation products. | High-resolution MS data is highly valuable for definitive identification. |
| Validated Stability-Indicating Assay | Official method for confirmatory and routine testing. | Must be cross-validated using forced degradation samples. |
| Chemical Reference Standards | For potential degradation products, when available. | Used to confirm identity and quantify specific degradants; demonstrates thoroughness. |
| Appropriate Solvents & Buffers | For forced degradation sample preparation. | Should represent a range of physiologically relevant conditions. |
| Primary Packaging Materials | For confirmatory testing of the drug product. | Testing in the final marketed package is mandatory for ICH Q1B. |
Photostability testing, mandated by ICH Q1B, is often viewed as a regulatory checkbox. However, this perspective undervalues its potential as a critical tool in formulation science. This whitepaper reframes photostability data from a confirmatory exercise into a proactive engine for developing robust drug products. By integrating forced degradation studies within a holistic development thesis, scientists can deconstruct degradation pathways, identify critical quality attributes, and design formulations that inherently resist photolytic stress, ultimately ensuring patient safety and product efficacy throughout the lifecycle.
The ICH Q1B guideline outlines two complementary approaches: confirmatory testing (standardized exposure to confirm package suitability) and forced degradation studies (extreme conditions to elucidate inherent stability). The broader thesis posits that forced degradation is not merely a stability-indicating method development tool but the cornerstone of a quality-by-design (QbD) formulation strategy. Data generated here informs every subsequent development decision, moving the paradigm "beyond compliance."
Strategic Integration of Forced Degradation in Development
A proactive photostability strategy aligns forced degradation studies with key development milestones.
Diagram Title: Proactive Photostability Strategy Flow
Quantitative Data from Photolytic Stress: A Comparative Analysis
The following table summarizes typical photodegradation outcomes for common functional groups, illustrating the quantitative foundation for formulation decisions.
Table 1: Common Photodegradation Pathways and Quantitative Degradation Ranges
| API Functional Group/Class | Primary Degradation Pathway | Typical % Degradation Observed in Forced Degradation* | Key Degradant Identified |
|---|---|---|---|
| Nitroaromatics | Reduction, ring rearrangement | 15-40% | Nitroso derivative, hydroxylamine |
| Tetracycles (e.g., Doxycycline) | N-demethylation, epoxy formation | 20-60% | 4-Epidoxycycline, lumidoxycycline |
| Quinolones (e.g., Ciprofloxacin) | Defluorination, piperazine ring modification | 10-30% | Desfluoroquinolone, ethylenediamine analog |
| Chlorpromazine (Phenothiazines) | Sulfoxide formation, ring cleavage | 25-70% | Chlorpromazine sulfoxide, promazine |
| Dihydropyridines (e.g., Nifedipine) | Photooxidation to pyridine derivative | 50-100% | Nitroso-pyridine analog |
| Retinoids (e.g., Tretinoin) | Isomerization, cyclization | 30-80% | cis-isomers, anhydrides |
*Conditions: ICH Option 2 (1.2 million lux hours, 200 W·h/m² UV), sample-dependent.
Detailed Experimental Protocol: Advanced Forced Degradation Study
This protocol extends beyond ICH Q1B minimums to generate formulation-relevant data.
Objective: To characterize the photodegradation kinetics, identify degradation products, and determine quantum yield of degradation for critical formulation screening.
Materials & Equipment:
- Drug Substance (Neat)
- Prototype Formulations (Solution, Suspension, Solid-Dispersion, Coated Tablet)
- SUNTEST CPS+ or equivalent calibrated light exposure system
- Controlled-temperature sample stage (±2°C)
- UV/VIS Spectrophotometer with integrating sphere
- HPLC-DAD-MS/MS system
- Narrow-bandpass filters (e.g., 313 nm, 420 nm)
- Chemical Actinometer (e.g., potassium ferrioxalate)
Procedure:
- Sample Preparation: Prepare thin-film samples of pure API and prototype formulations on quartz plates. For solid oral doses, use a powder bed or intact dosage form.
- Actinometry: Calibrate the light source intensity (E, W/m²) at specific wavelengths using the chemical actinometer in parallel with sample exposure.
- Exposure Matrix: Expose samples in a time-series (e.g., 0, 2, 6, 12, 24, 48 hours) under:
- Full ICH spectrum (UV + Visible).
- Filtered UV only (λ ≤ 400 nm).
- Filtered Visible only (λ ≥ 400 nm).
- Kinetic Analysis: At each interval, quantify remaining parent compound via validated HPLC. Plot Ln(%Remaining) vs. Radiant Exposure (H, J/m²). The slope = apparent degradation rate constant (k).
- Quantum Yield Calculation: For key wavelengths, calculate the quantum yield of degradation (Φdeg) using: Φdeg = (Rate of molecule degradation) / (Rate of photon absorption). Rate of photon absorption derived from actinometry and sample absorbance.
- Degradant Mapping: Isolate and identify major degradants (>0.1%) from each formulation type using HPLC-MS/MS and NMR.
Diagram Title: Advanced Photodegradation Study Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Photostability-Driven Formulation Research
| Item/Category | Function & Rationale |
|---|---|
| Chemical Actinometers (e.g., Potassium Ferrioxalate, Aberchrome 670) | Precisely measures photon flux (light intensity) at specific wavelengths. Critical for calculating quantum yield and comparing studies across laboratories. |
| Monochromatic Light Filters/Narrow-Band LEDs | Isolates specific wavelengths of light (e.g., 305 nm, 365 nm, 450 nm) to determine the API's action spectrum and match protective strategies to the most damaging wavelengths. |
| Transparent Inert Substrates (Quartz, Suprasil Slides) | Holds thin-film samples without absorbing UV light, ensuring accurate and reproducible light exposure for kinetic studies. |
| Radical Scavengers & Quenchers (e.g., Sodium Azide, Mannitol, β-Carotene) | Probes degradation mechanisms (e.g., singlet oxygen vs. free radical) when added to solution formulations, guiding excipient selection. |
| Spectroscopic Grade Solvents | Ensures no UV-absorbing impurities interfere with sensitive photochemical reactions or analytical quantification. |
| Polymer Film Coating Systems (e.g., OPADRY II UV White) | Prototype coatings with varying levels of UV absorbers (TiO₂, iron oxides) to test physical light barrier efficacy on solid dosage forms. |
| Validated HPLC-DAD-MS/MS System | The core analytical tool for simultaneous quantification of parent compound loss and structural elucidation of photodegradants. |
Translating Data into Robust Formulation Design
The mechanistic insights guide specific formulation interventions:
- If degradation is UV-driven (λ < 400 nm): Opaque packaging (amber glass, aluminum blisters) is highly effective. TiO₂ or iron oxide coatings on tablets provide robust internal protection.
- If degradation is visible-light driven (λ ≥ 400 nm): Amber packaging may be insufficient. Formulation-based quenching using approved dyes or antioxidants becomes critical.
- If Quantum Yield (Φ_deg) is high (>0.01): The molecule is highly photosensitive. Aggressive protective strategies in both formulation and packaging are required.
- If degradation occurs via singlet oxygen: Incorporate singlet oxygen quenchers (e.g., α-tocopherol) or use antioxidants that compete for reactive oxygen species.
Table 3: Formulation Strategy Matrix Based on Photostability Data
| Photostability Profile | Primary Strategy | Secondary Strategy | Packaging Implication |
|---|---|---|---|
| High Φ_deg, UV-specific | High-load TiO₂ film coating | Antioxidant in core | Amber glass required |
| Moderate Φ_deg, Broad-spectrum | Combination coating (TiO₂ + FeO) | Sacrificial agent in matrix | Opaque or light-resistant blister |
| Low Φ_deg, Visible-light specific | Molecular encapsulation (Cyclodextrins) | Selective dye/quencher | May require non-standard coloring |
Treating photostability testing as a foundational, knowledge-generating exercise transforms it from a cost center into a value driver. The quantitative kinetics, mechanistic pathways, and quantum yield data derived from well-designed forced degradation studies provide an unprecedentedly clear roadmap for formulating intrinsically stable products. This proactive approach, grounded in the broader thesis of ICH Q1B's dual purpose, mitigates late-stage failures, accelerates development, and ultimately delivers more reliable medicines to patients. The goal is not just to pass a test, but to build a product where the test is merely a formality.
Conclusion
Confirmatory testing under ICH Q1B and forced degradation studies are not interchangeable but are fundamentally complementary. Confirmatory studies provide the definitive, standardized evidence of photostability required for market authorization, while forced degradation is an indispensable development tool that reveals vulnerability and mechanistic pathways. A strategic integration of both approaches enables robust formulation design, scientifically justified packaging selection, and proactive risk mitigation. The future of photostability testing lies in advancing analytical methodologies for degradant identification and potentially refining stress conditions to better predict real-world stability, ultimately strengthening the overall quality and safety profile of pharmaceutical products throughout their lifecycle.